Keeping the ions close: A new activity

Improving chemical reactions ranging from refining oil to building longer-lasting batteries means understanding the chemistry of acids and bases. Researchers discovered that when a strong acid such as hydrochloric acid (HCl) is mixed with water, the negatively charged anion and positively charged cation remain close and create an unexpected structure. These results provide a better understanding of the complexity of acid/base chemistry in concentrated, non-ideal chemical solutions.
This new study – detailing the important role of the counter ion (chloride in the case of HCl) in the reaction network of acids behavior – provides a template to connect structure and function. The connection can help scientists develop a deeper understanding of the most basic concepts in chemistry and provide foundational information for use in battery applications.
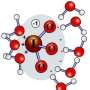
Introductory chemistry textbooks state that when an acid is added to water that the positive and negatively charged ions quickly separate from each other based on research with low acidic concentrations. With strong acids, such as hydrochloric acid (HCl), it was thought to be the same situation: the positive ions of hydrogen (H+) diffuse – creating the acidity of the solution, neutralizing basic substances, corroding metals and/or reacting with organic substances – and then quickly associate with water to become a hydronium ion or H3O+, while the chloride counter ion (CL-) forms independent, negatively charged solvated fragments. However, the fundamental properties of the counter ions surrounded by water have not been thoroughly experimentally studied. Now researchers using computational and experimental methods examine low, medium, and high concentrations of HCl in liquid water and discovered that the negatively charged CL- and the positively charged H+ actually remain closer to each other in an unexpected structure throughout the entire concentration range studied.
The researchers at Pacific Northwest National Laboratory combined extended X-ray absorption fine structure, neutron diffraction, and X-ray diffraction measurements using the Argonne Advanced Photon Source with state-of-the-art density functional theory simulations. Specifically, the researchers measured the distance and geometry between the chloride ion and the oxygen in the hydronium ion comprising the contact ion pair – finding that it was significantly shorter than the interaction between chloride and the oxygen of water as expected in an ideal dissociation picture. These structures differed significantly from those studied in the gas phases, which were used in prior computational models. The team is now examining more complex ions with the idea of tuning them to specific purposes.
More information: "Persistent ion pairing in aqueous hydrochloric acid." The Journal of Physical Chemistry B 118, 7211 (2014). DOI: 10.1021/jp501091h
Journal information: Journal of Physical Chemistry B
Provided by US Department of Energy