Aluminum could give a big boost to capacity and power of lithium-ion batteries
One big problem faced by electrodes in rechargeable batteries, as they go through repeated cycles of charging and discharging, is that they must expand and shrink during each cycle—sometimes doubling in volume, and then shrinking back. This can lead to repeated shedding and reformation of its "skin" layer that consumes lithium irreversibly, degrading the battery's performance over time.
Now a team of researchers at MIT and Tsinghua University in China has found a novel way around that problem: creating an electrode made of nanoparticles with a solid shell, and a "yolk" inside that can change size again and again without affecting the shell. The innovation could drastically improve cycle life, the team says, and provide a dramatic boost in the battery's capacity and power.
The new findings, which use aluminum as the key material for the lithium-ion battery's negative electrode, or anode, are reported in the journal Nature Communications, in a paper by MIT professor Ju Li and six others. The use of nanoparticles with an aluminum yolk and a titanium dioxide shell has proven to be "the high-rate champion among high-capacity anodes," the team reports.
Most present lithium-ion batteries—the most widely used form of rechargeable batteries—use anodes made of graphite, a form of carbon. Graphite has a charge storage capacity of 0.35 ampere-hours per gram (Ah/g); for many years, researchers have explored other options that would provide greater energy storage for a given weight. Lithium metal, for example, can store about 10 times as much energy per gram, but is extremely dangerous, capable of short-circuiting or even catching fire. Silicon and tin have very high capacity, but the capacity drops at high charging and discharging rates.
Aluminum is a low-cost option with theoretical capacity of 2 Ah/g. But aluminum and other high-capacity materials, Li says, "expand a lot when they get to high capacity, when they absorb lithium. And then they shrink, when releasing lithium."
This expansion and contraction of aluminum particles generates great mechanical stress, which can cause electrical contacts to disconnect. Also, the liquid electrolyte in contact with aluminum will always decompose at the required charge/discharge voltages, forming a skin called solid-electrolyte interphase (SEI) layer, which would be ok if not for the repeated large volume expansion and shrinkage that cause SEI particles to shed. As a result, previous attempts to develop an aluminum electrode for lithium-ion batteries had failed.
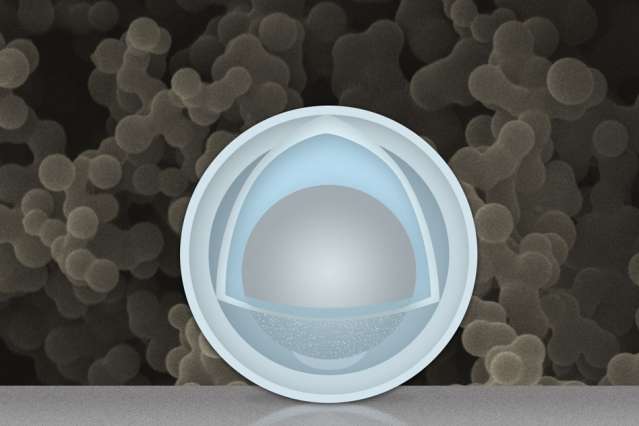
That's where the idea of using confined aluminum in the form of a yolk-shell nanoparticle came in. In the nanotechnology business, there is a big difference between what are called "core-shell" and "yolk-shell" nanoparticles. The former have a shell that is bonded directly to the core, but yolk-shell particles feature a void between the two—equivalent to where the white of an egg would be. As a result, the "yolk" material can expand and contract freely, with little effect on the dimensions and stability of the "shell."
"We made a titanium oxide shell," Li says, "that separates the aluminum from the liquid electrolyte" between the battery's two electrodes. The shell does not expand or shrink much, he says, so the SEI coating on the shell is very stable and does not fall off, and the aluminum inside is protected from direct contact with the electrolyte.
The team didn't originally plan it that way, says Li, the Battelle Energy Alliance Professor in Nuclear Science and Engineering, who has a joint appointment in MIT's Department of Materials Science and Engineering.
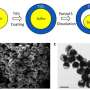
"We came up with the method serendipitously, it was a chance discovery," he says. The aluminum particles they used, which are about 50 nanometers in diameter, naturally have an oxidized layer of alumina (Al2O3). "We needed to get rid of it, because it's not good for electrical conductivity," Li says.
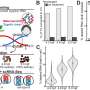
They ended up converting the alumina layer to titania (TiO2), a better conductor of electrons and lithium ions when it is very thin. Aluminum powders were placed in sulfuric acid saturated with titanium oxysulfate. When the alumina reacts with sulfuric acid, excess water is released which reacts with titanium oxysulfate to form a solid shell of titanium hydroxide with a thickness of 3 to 4 nanometers. What is surprising is that while this solid shell forms nearly instantaneously, if the particles stay in the acid for a few more hours, the aluminum core continuously shrinks to become a 30-nm-across "yolk,",which shows that small ions can get through the shell.
The particles are then treated to get the final aluminum-titania (ATO) yolk-shell particles. After being tested through 500 charging-discharging cycles, the titania shell gets a bit thicker, Li says, but the inside of the electrode remains clean with no buildup of the SEIs, proving the shell fully encloses the aluminum while allowing lithium ions and electrons to get in and out. The result is an electrode that gives more than three times the capacity of graphite (1.2 Ah/g) at a normal charging rate, Li says. At very fast charging rates (six minutes to full charge), the capacity is still 0.66 Ah/g after 500 cycles.
The materials are inexpensive, and the manufacturing method could be simple and easily scalable, Li says. For applications that require a high power- and energy-density battery, he says, "It's probably the best anode material available." Full cell tests using lithium iron phosphate as cathode have been successful, indicating ATO is quite close to being ready for real applications.
"These yolk-shell particles show very impressive performance in lab-scale testing," says David Lou, an associate professor of chemical and biomolecular engineering at Nanyang Technological University in Singapore, who was not involved in this work. "To me, the most attractive point of this work is that the process appears simple and scalable."
There is much work in the battery field that uses "complicated synthesis with sophisticated facilities," Lou adds, but such systems "are unlikely to have impact for real batteries. … Simple things make real impact in the battery field."
More information: "High-rate aluminium yolk-shell nanoparticle anode for Li-ion battery with long cycle life and ultrahigh capacity." Nature Communications 6, Article number: 7872 DOI: 10.1038/ncomms8872
Journal information: Nature Communications
Provided by Massachusetts Institute of Technology
This story is republished courtesy of MIT News (web.mit.edu/newsoffice/), a popular site that covers news about MIT research, innovation and teaching.