Together bacteria invade antibiotic landscapes
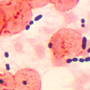
Antibiotics kill bacteria – or at least they are supposed to, although unfortunately this does not always result in a cure. Scientists at TU Delft's Kavli Institute of Nanoscience have discovered that bacteria can colonise environments in which there are deadly quantities of antibiotics, without becoming genetically resistant. In order to achieve this, the bacteria need to work together in large groups, as individually they would not stand a chance. The research will help scientists to improve their understanding of how bacteria survive when there are local variations in antibiotic concentrations – a situation which occurs in your body when you take an antibiotic tablet. These new insights will enable scientists to understand why antibiotics sometimes fail, and provide cues to improve their effectiveness.
From 'heaven' to 'hell'
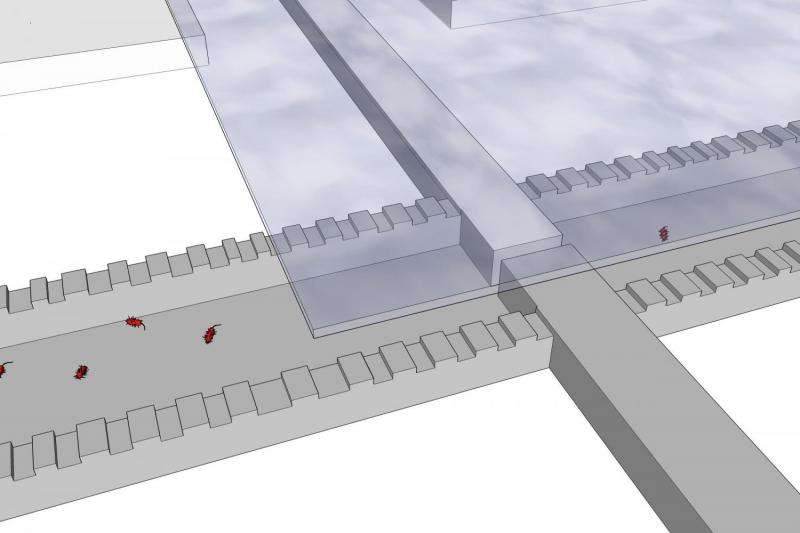
For the research, biophysicist Felix Hol worked with researchers Bert Hubert, Cees Dekker and Juan Keymer to build two chambers approximately 8mm in length, linked together by a very narrow connector through which bacteria can swim. "One chamber is a paradise for bacteria, with a constant flow of nutrients, where they can multiply very rapidly", explains Hol. "The other chamber is a proverbial hell: in addition to nutrients, it contains a very high dose of the antibiotic kanamycin, which is lethal for these bacteria. Although you might expect the bacteria to keep well away from it, it turns out that they do not. Our research shows that the bacteria join forces in an attempt to colonise the deadly chamber. This is only successful when extremely dense groups of bacteria invade the chamber."
Large groups
However, not every 'invasion' is successful. "Initially, the population density in the 'heaven' chamber is still quite low. The first groups that make the crossing to the 'hell' chamber are often still quite small, consisting of up to 100 bacteria, and they do not survive. After approximately eight hours, the groups become so large and dense that they can successfully colonise the deadly environment. The bacteria then multiply in the presence of the antibiotic and the population in the deadly environment increases to the same size as those in the bacteria paradise," says Felix Hol. "Colonisation is not always successful: In four of the ten experiments, the bacteria succeeded in colonising the compartment containing antibiotics, while in two they did not. In the remaining four cases the bacteria did invade the compartment, yet the population did not survive."
No resistance
What is striking is that the bacteria did not develop resistance to the antibiotic after colonising the deadly environment. "When I examined the bacteria afterwards, it turned out that, with the exception of a single experiment, the bacteria had not become resistant to the antibiotic. They are able to work together to create an effective defense by adapting collectively in one way or another." The research could improve understanding of the way bacteria behave in the human body. In the body, drugs like antibiotics form concentration gradients: for example, the concentration is much higher in the intestines than in the lungs, but major differences can also develop because drugs are taken periodically. "The research demonstrates how bacteria deal with these kinds of gradients. Ultimately, this can improve our understanding of the relative efficacy of certain drugs and why some bacterial infections are difficult to cure", says Professor of Bionanoscience Cees Dekker. "It can also help us to gain a better understanding of how bacteria become resistant to drugs. It could be that this form of colonisation which requires no resistance is actually an initial step, after which resistant bacteria develop in the deadly environment through natural selection."
Unexplained mechanism
The precise mechanism by which bacteria are able to colonise the deadly environment remains largely unexplained. One hypothesis is that they behave in a similar way to a 'Testudo', the tortoise-like formation adopted by Roman soldiers in which they join forces to create a large shield that forms an effective buffer in a hostile environment. In follow-up research, the scientists hope to unravel the exact biological mechanism that enables the bacteria to exhibit this behaviour.
More information: Density-dependent adaptive resistance allows swimming bacteria to colonize an antibiotic gradient, The ISME Journal advance online publication, 3 July 2015; DOI: 10.1038/ismej.2015.107
Journal information: ISME Journal
Provided by Delft University of Technology