Researchers rapidly finding new applications for coherent diffractive imaging
In 1999, UCLA professor John Miao pioneered a technique called coherent diffractive imaging, or CDI, which allows scientists to re-create the 3D structure of noncrystalline samples or nanocrystals. The achievement was extremely significant because although X-ray crystallography had long allowed scientists to determine the atomic structure of a wide variety of molecules, including DNA, it does not work for noncrystalline materials used in a variety of disciplines, including physics, chemistry, materials science, nanoscience, geology and biology.
An article by Miao and his colleagues in the latest issue of Science reviews and analyzes the rapid development of brilliant X-ray sources that scientists worldwide have used for a broad range of applications of his invention in physical and biological sciences.
CDI now is being used in a wider array of applications than Miao had imagined it would be—and the technique has become ever more important to scientists exploring the borders of observable nanoscience.
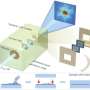
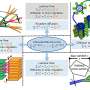
Miao, a professor of physics and astronomy, found that by illuminating a noncrystalline sample with a brilliant laserlike, or coherent, X-ray, he could use a lensless detector to record the pattern, or diffraction, of the scattering X-rays. He then recreated the 3D structure of the sample by developing advanced phase retrieval algorithms applied to the diffraction pattern, which is why his technique is sometimes referred to as lensless imaging.
CDI transformed the conventional view of microscopy by replacing the physical lens with a computational algorithm. By avoiding the use of lenses, CDI can obtain images of nanoscale objects with high resolution and high contrast. It also has advantages over other imaging techniques such as electron microscopy because it can be used to image thick samples in three dimensions.
This powerful imaging technique is now expected to profoundly expand our understanding of a wide range of dynamic phenomena in physics, chemistry and microelectronics; for example, phase transitions, when substances change quickly from one state to another.
CDI is ideal for quantitative 3D characterization of nanoscale materials for several reasons. X-rays have a larger penetration depth than electrons, so samples in an electron microscope are destroyed by the powerful electron beam of the microscope as they are imaged, but CDI's X-rays can often avoid sample destruction. CDI also enables nanoscale chemical, elemental, and magnetic 3D mapping of complex matter.
In materials science, CDI was used to determine the first 3D deformation field and full strain tensor inside individual nanocrystals with nanoscale resolution, a key to understanding and managing strain, which is fundamental to designing and implementing nanomaterials such as those used in high-speed electronics. CDI also made possible the first 3D imaging of mineral crystals inside bones at the nanometer scale, giving a much greater understanding of the molecular structure of bone.
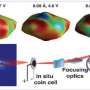
In lithium ion batteries, when the electrode material stores electrical charge, the material undergoes phase transition that reduces the battery's life. With CDI, scientists can better understand how lithium ion batteries can be made to store more energy and last longer without cracking.
More information: "Beyond Crystallography: Diffractive Imaging Using Coherent X-ray Light Sources." Science 1 May 2015: Vol. 348 no. 6234 pp. 530-535 DOI: 10.1126/science.aaa1394
Journal information: Science
Provided by University of California, Los Angeles