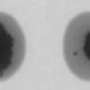
'Explosive' atom movement is new window into growing metal nanostructures
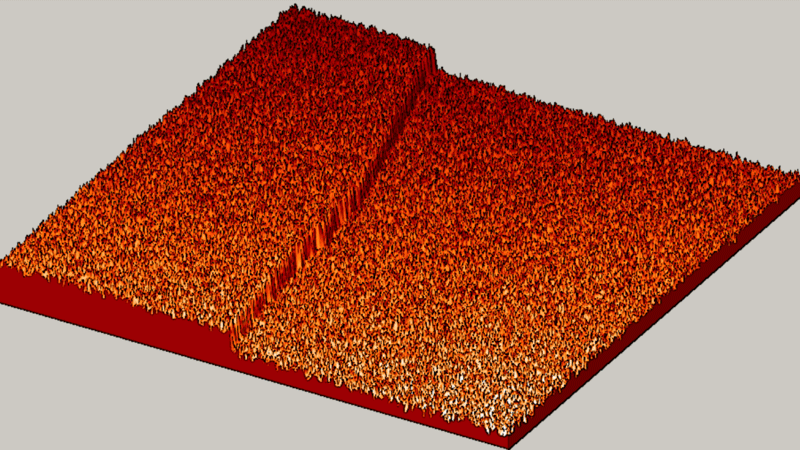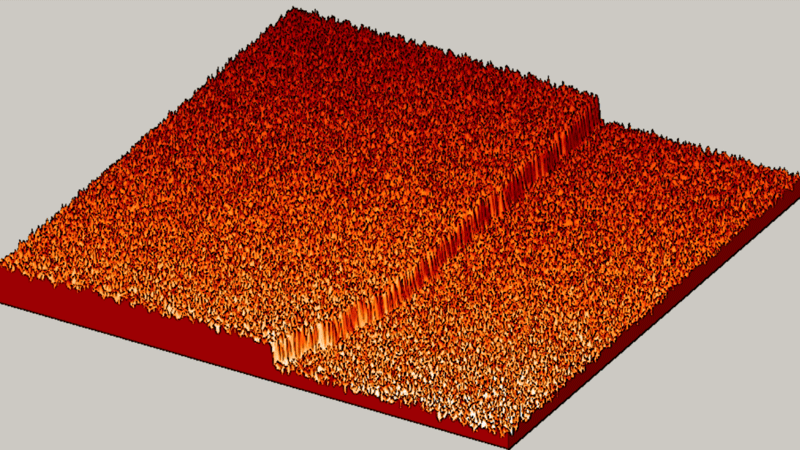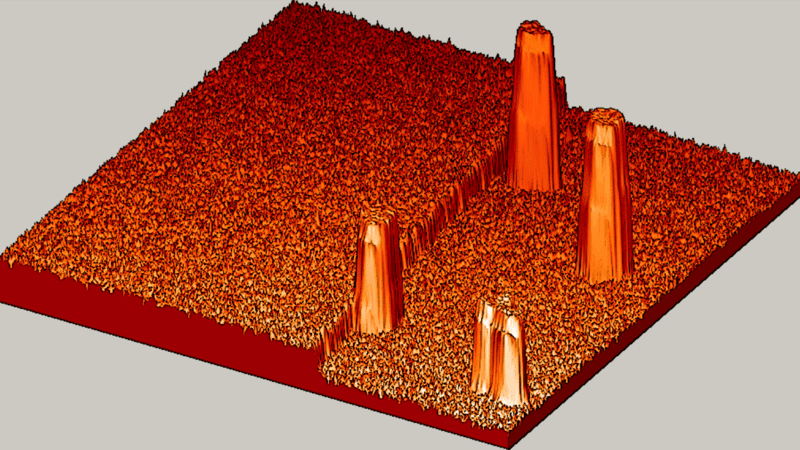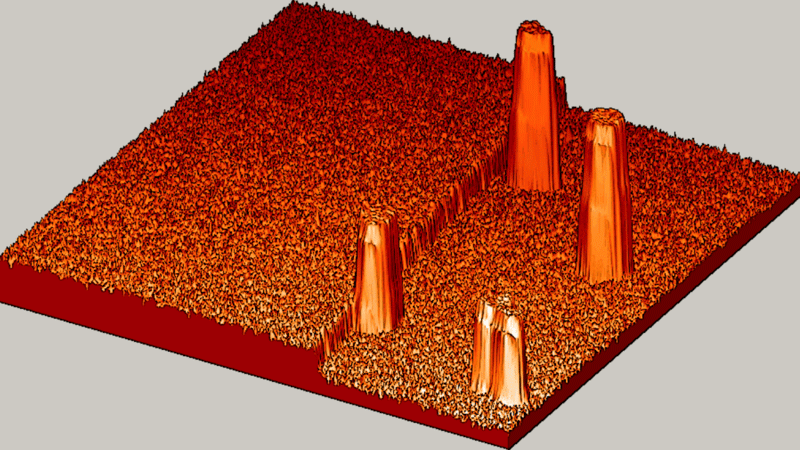
"The textbook said we should see slow, gradual and random. But what we saw? BOOM! Fast, explosive and organized!" said Michael Tringides, physicist at the U.S. Department of Energy's Ames Laboratory and a professor of physics and astronomy at Iowa State University.
Tringides is talking about the unusual atom movement he saw when they dropped a few thousand lead atoms onto a flat, smooth lead-on-silicon surface, all at low temperatures, and looked at an area just one-twentieth the width of a human hair.
What the Ames Laboratory scientists expected to see was "random-walk diffusion": atoms milling around, looking like they have no idea where they're going, where they've been, or that any fellow atoms are near them. Typically, the atoms eventually happen to run into each other and create small structures.
"Instead, we saw atoms that are very focused and work together well to quickly create tiny lead nanostructures," said Tringides. "That kind of 'collective diffusion,' is really the exception to the rule in atom movement. Plus, we were surprised by how fast well-organized crystal structures nucleate at such cold temperatures, where movement is typically slow."
The collective, fast diffusion observed by Tringides' team could represent a new way to grow perfect, tiny metal nanostructures.
"If we're able to make a nanoscale lead object this fast, we can perhaps create other objects this way." said Tringides. "Understanding the basic science of how materials work at these nanoscales may be key to making nanotransistors, nanoswitches and nanomagnets smaller, faster and reliably."
Tringides' research team specializes in measuring how atoms move on surfaces, revealing through scanning tunneling microscopy how the smallest structures begin to form. Over the past several years, they've used their expertise to answer fundamental questions about materials, such as rare-earths, graphene and metallic films, that are important to green energy technologies.
This research, which appeared in Physical Review Letters, is supported by the U.S. Department of Energy Office of Science.
More information: Hershberger, M. T., Hupalo, M., Thiel, P. A., Wang, C. Z., Ho, K. M., & Tringides, M. C. (2014). Nonclassical "Explosive" Nucleation in Pb/Si(111) at Low Temperatures. Physical Review Letters, 113(23). DOI: 10.1103/physrevlett.113.236101
Journal information: Physical Review Letters
Provided by Ames Laboratory