January 20, 2015 report
Sonic booms in nerves and lipid membranes

(Phys.org)—Neurons might not be able to send signals as fast as electrons in wires or photons in fiber, but what if they can communicate using miniature sonic booms? That would be quite a revolutionary discovery. A group of mechanical engineers at Boston University have recently found that two-dimensional shock waves reminiscent of propagating spikes in nerves can be induced in monolayer lipid membranes. Their fascinating results, which have been sitting on the Arxiv server since November, have just been approved for publication in an upcoming issue of Physical Review E.
The idea that action potentials in neurons are acoustic pressure pulses has gained a lot of experimental support. Shamit Shrivastava and Matthias Schneider were previously able to directly observe solitary waves in membranes using an optical technique known as fluorescence resonance energy transfer, or FRET. Following that, Thomas Heimburg's group demonstrated that counter-propagating action potentials don't annhilate in nerves in they way they would be expected to if they were purely electrical spikes possessing refractory periods and channel inactivation. In work that is largely complimentary to these shock studies, Heimburg has shown that the pulses survive collision and continue to travel in their opposite directions.
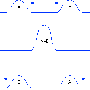
Although solitary waves and compressive shock waves (and for that matter expansive ones too) are not mutually exculsive, shock waves generally differ in that their energy quickly dissipates with distance. A unique feature of a shock disturbance is it can travel significantly faster than an ordinary wave in a given media. To demonstrate that these effects exist in lipid monolayers, Shrivastava and Schneider (together with Kevin Heeyong Kang) varied the amplitude of the shock compression and measured the velocity of the propagating wave in a special setup known as Langmuir trough. They report a relationship between interface pressure and density under adiabatic conditions—conditions where there is no heat exchange—which yields an adiabatic state diagram.
Shivastrava mentioned that the general literature on shock and detonation may shed light on the interaction of chemistry with sound waves. In particular he suggests ion channels might actually feed a sound wave and compensate for the dissipation that has to be expected from propagation. This conception bears some resemblance to other ideas recently proposed regarding the natural decay of pressure pulses in the viscoelastic medium of the nerve. According to one hypothesis, pulses in myelinated nerve segments would be mechanically refreshed by ionic input at the nodes, while the muscle end-plate also participates in the pulse after amplification by acetylcholine release in the cleft. One reason why many ion channels also appear to be mechanosensitive may in fact be because of coordinated lipid motions on the relevant scales.
State diagrams and thermodynamic relationships may at first seem to be a bit removed from the everyday biophysical world of spikes in protein-rich bilayers of neurons. However, we should not fear to tread further. The particular form of the state diagram determines many critical parameters of excitations in lipid membranes including the shape, stability, and velocity. While the pressures and temperatures relevant to lipids may be different to those in sonic booms generated by supersonic aircraft, much of the underlying physics is the same.
Having this physics base distinguishes these experiments from other kinds of claims of percussive booms in neurons which one may have recently heard about. Here I am speaking particularly about the kinds of 'brain shocks' hypothesized to occur from the self-propagating decomposition of peroxynitrate compounds in the brain or sensory organs. In this case, a small detonation pulse producing charged betaines would apparently liberate heat with subsequent pressure increases and local gasification. Shivastava's work we should mention, would actually predict that liberation of heat near phase transitions can actually lead to condensation in lipids.
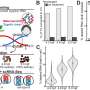
In contrast to this kind of fantastical speculation about betaine detonation, the thermodynamic measurements now being made in neurons have a deep history. Many of the ideas currently being explored can be traced to the work of Konrad Kaufmann, who among other pursuits developed a theory of proton action at cholinergic synapses (e.g. neuromuscular junctions). Some of his early propositions of acetlycholinesterase being an enzymatic 'proton pistol' (which led to later ideas about the thermodynamics of nerves) are now being confirmed experimentally. In another paper on the Arxiv server, Schneider and Christian Fillafer demonstrate membrane depolarization and spike generation by hydrolyzed acetylcholine.
In other words, we now have another heresy here implying that it is not so much ACh's action on receptor proteins that generates membrane activity, rather, the rapid breakdown of ACh by acetylcholinesterases releases protons which then excite the postsynaptic membrane. These new experiments were not done in neurons, or even in the often used frog oocytes since there are several complicating variables at play. For instance, oocytes have intrinsic esterase activity as well as ACh-sensitivity which in practice needs to be supressed by addition of inhibitors like atropine. These acetylcholinesterase can actually be found in a lot of places one might not expect them. They are in the eggs and sperm, the placenta and early differentiating embryo, in red blood cells in some organisms, and directly in the blood of others.
Instead of the usual suspects, here the researchers used excitable Chara australis internodal cells—a plant cell which has a long history of use as a model system in electrophysiological experiments. While action potentials were observed in frog preparations as early as the nineteenth century, the first intracellular electrode recordings of action potentials were done in Chara cells—some ten years before the famous squid axon recordings. This general concept of proton excitation may extend to other transmitters like excitatory amino acids, or even to bases like serotonin. One might, for example, want to investigate whether glutamate is deprotonated in the synaptic cleft. The established mechanisms taking place in the presynapse, where the pH-dependant uptake of glutamate into acidic vessicles happens, may or may not support instances of glutamate being released in protonated form.
In talking to Shivastava and to Fillafer, I get the feeling that we should expect a lot of revolutionary new work coming out of their lab. We will have more to report here soon about these cutting edge experiments.
More information: — Solitary Shock Waves and Adiabatic Phase Transition in Lipid Interfaces and Nerves, arxiv.org/pdf/1411.2454v3.pdf
— On the excitation of action potentials by protons and its potential implications for cholinergic transmission, arxiv.org/pdf/1411.7912v1.pdf
Journal information: arXiv , Physical Review E
© 2015 Phys.org