January 27, 2015 report
Quantum dots combined with antibodies as a method for studying cells in their native environment
(Phys.org)—To understand cell function, we need to be able to study them in their native environment, in vivo. While there are many techniques for studying cells in vitro, or in the laboratory setting, in vivo studies are much more difficult. A new study by a team of researchers at the Massachusetts Institute of Technology and Harvard Medical School used a unique quantum dot-antibody conjugate to facilitate in vivo studies of bone marrow stem cells in mice. This study was reported in the Proceedings of the National Academy of Science.
Typically, to study a cell in vivo involves making invasive modifications to the cell or the organism that disrupt the cell's native environment. Additionally, many in vivo studies involve studying groups of cells, rather than tracking a single cell. Prior techniques involved manipulating the cells by immunohistochemistry, genetic engineering, or irradiation of the organism. All of these techniques either create substantial changes to the native environment, or they are only able to look at a "snapshot" of the cell interacting with its environment. It cannot study the movement of the cell throughout the body.
Quantum dots are semi-conductor-like nanoparticles with optical properties that can be finely tuned for a wide range of optical-based studies, including infrared and fluorescence. Han, et al. targeted a particular cell type by combining quantum dots with antibodies matched to the cell's surface receptors, so that they would combine like a lock and key .
Their quantum dot-antibody system was built from quantum dots combined with polyimidazole ligands (PILs) and norbornene. PILs are highly stable and will coat the surface of quantum dots. Norbornene is a versatile functional group that maintains a neutral charge, making it a good choice for diffusing throughout the body. Norbornene was attached to an antibody that was specific for Sca1+c-Kit+ cells, which are a type of stem cell found in the calvarial bone marrow.
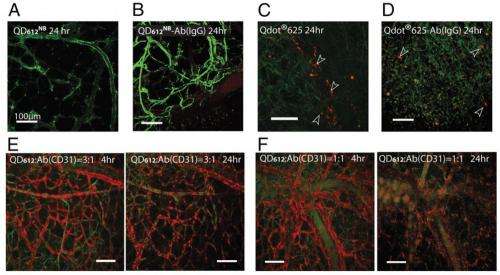
The quantum dot-antibody conjugates were small enough to diffuse through the cell and were specific enough that they did not attach to unwanted cells. Additionally, they provided an adequate signal for optical studies and flow cytometry, allowing the study of Sca1+c-Kit+ cell diffusion in the bone marrow of unmanipulated mice.
This method for studying single cells in their native environment is versatile enough to be used for other cell types by attaching different antibodies to a quantum dot. Additionally, the study showed that the quantum dot-antibody conjugates were highly stable with a long circulation half-life, allowing for a more extensive study of cellular interactions in vivo. Finally, the purification process produced highly pure conjugates with few unbound molecules, and the quantum dot-antibody conjugate size was appropriate for diffusion through the mouse. This research has broader applications, as many of the factors the researchers addressed are constraints for any in vivo cell studies.
More information: Quantum dot/antibody conjugates for in vivo cytometric imaging in mice, Hee-Sun Han, PNAS, DOI: 10.1073/pnas.1421632111
Abstract
Multiplexed, phenotypic, intravital cytometric imaging requires novel fluorophore conjugates that have an appropriate size for long circulation and diffusion and show virtually no nonspecific binding to cells/serum while binding to cells of interest with high specificity. In addition, these conjugates must be stable and maintain a high quantum yield in the in vivo environments. Here, we show that this can be achieved using compact (∼15 nm in hydrodynamic diameter) and biocompatible quantum dot (QD) -Ab conjugates. We developed these conjugates by coupling whole mAbs to QDs coated with norbornene-displaying polyimidazole ligands using tetrazine–norbornene cycloaddition. Our QD immunoconstructs were used for in vivo single-cell labeling in bone marrow. The intravital imaging studies using a chronic calvarial bone window showed that our QD-Ab conjugates diffuse into the entire bone marrow and efficiently label single cells belonging to rare populations of hematopoietic stem and progenitor cells (Sca1+c-Kit+ cells). This in vivo cytometric technique may be useful in a wide range of structural and functional imaging to study the interactions between cells and between a cell and its environment in intact and diseased tissues.
Journal information: Proceedings of the National Academy of Sciences
© 2015 Phys.org