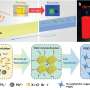
Nanotubes could serve as 'universal scaffolding' for cell membrane channels
A study, in which the Membrane Nanomechanics group led by the Ikerbasque lecturer Dr. Vadim Frolov at the Biophysics Unit of the University of the Basque Country has participated, suggests that single-wall carbon nanotubes could be used as universal scaffolding to help to replicate the properties of cell membrane channels. The results of the study have been published in the prestigious journal Nature.
Biological membranes define the functional architecture of living systems: they are selectively permeable, maintain the chemical identity of the cells and intracellular organelles, and regulate the exchange of material between them. To control the transporting of ions and small molecules through cell membranes, highly specialised proteins that transport these molecules through the membrane are used. Recent advances in nanotechnology and nanofabrication have made it possible to synthesise and manufacture artificial compounds destined to fulfil the functions of transmembrane channels and transporters. The behaviour of these artificial compounds is increasingly similar to that of their cell prototypes, in other words, they have increasingly similar characteristics: molecular selectivity, membrane targeting and transport efficiency. However, creating a universal, versatile prototype to manufacture channels with specific transport properties remains a challenge.
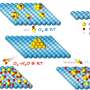
The study, which has had the participation of the group of Dr Vadim Frolov, Ikerbasque lecturer-researcher at the UPV/EHU's Biophysics Unit, and led by Dr Alex Noy of the Lawrence Livermore National Laboratories (United States), suggests that single-wall carbon nanotubes (CNTs) can be used as a framework with similar affinity and transport properties as protein channels. Nanotubes are very efficient transporters because their narrow diameter (of about 1 nm) and hydrophobic interior are very similar to the general structural design of these proteins.
The researchers involved in the study have discovered that ultrashort CNTs covered with lipid molecules form channels in artificial membranes as well as in living cell membranes. These structures remain stable in solution and spontaneously insert into the membranes. Likewise, the researchers have seen that the CNTs inserted in a membrane contain transport properties comparable with those of small ion channels. What is more, they have found that these CNTs are capable of transporting DNA.
A Promising Future
As Frolov concludes, transmembrane transport mechanisms by means of ultrashort CNTs require more extensive research, so the collaboration project between the groups at the Lawrence Livermore National Laboratories and the UPV/EHU has not finished yet. The scientists are hoping that by means of sophisticated chemical modifications, the optimizing of the production processes, and the use of other nanofabrication approaches can succeed in producing fully functional ionic channels based on ultrashort CNTs.
More information: J. Geng, K. Kim, J. Zhang, A. Escalada, R. Tunuguntla, Luis R. Comolli, Frances I. Allen, Anna V. Shnyrova, Kang Rae Cho, D. Munoz, Y. MorrisWang, Costas P. Grigoropoulos, Caroline M. Ajo-Franklin, Vadim A. Frolov & A. Noy. Stochastic transport through carbon nanotubes in lipid bilayers and live cell membranes. Nature, 30 october 2014, Vol. 514. DOI: 10.1038/nature13817
Journal information: Nature
Provided by University of the Basque Country