A new approach to treating peanut and other food allergies
These days, more and more people seem to have food allergies, which can sometimes have life-threatening consequences. In ACS' Journal of Agricultural and Food Chemistry, scientists report the development of a new type of flour that someday could be used in food-based therapies to help people better tolerate their allergy triggers, including peanuts.
Mary Ann Lila and colleagues note that of the 170 foods that cause allergic reactions, peanuts can be the most dangerous. These reactions can range from mild itching and hives to life-threatening anaphylactic shock, in which a person's throat swells, making it difficult or impossible to breathe. An experimental treatment that involves giving minute quantities of the trigger food to patients over a period of time in a clinic is successful for some patients who are allergic to peanuts. The process, called desensitization, sets off beneficial responses by the body to the food. But the milled roasted peanut flour that is currently used can have severe side effects. Lila's team set out to design a new type of flour that could help control food allergies without causing dangerous side effects.
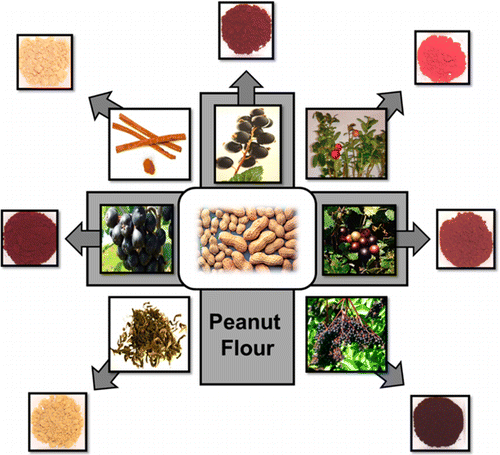
They turned to plant polyphenols, which have shown promise as compounds that can dampen allergic reactions. The scientists developed a modified flour powder in which cranberry polyphenols were bound to peanut proteins. With this extra cargo, the peanut-containing powder triggered the beneficial desensitization reactions, without provoking harmful allergic responses in laboratory tests with mice. The scientists note that the technique could also be adapted for other food allergies.
More information: "Novel Strategy To Create Hypoallergenic Peanut Protein-Polyphenol Edible Matrices for Oral Immunotherapy" J. Agric. Food Chem., Article ASAP. DOI: 10.1021/jf405773b
Abstract
Peanut allergy is an IgE-mediated hypersensitivity. Upon peanut consumption by an allergic individual, epitopes on peanut proteins bind and cross-link peanut-specific IgE on mast cell and basophil surfaces triggering the cells to release inflammatory mediators responsible for allergic reactions. Polyphenolic phytochemicals have high affinity to bind proteins and form soluble and insoluble complexes with unique functionality. This study investigated the allergenicity of polyphenol-fortified peanut matrices prepared by complexing various polyphenol-rich plant juices and extracts with peanut flour. Polyphenol-fortified peanut matrices reduced IgE binding to one or more peanut allergens (Ara h 1, Ara h 2, Ara h 3, and Ara h 6). Attenuated total reflectance−Fourier transform infrared spectroscopy (ATR-FTIR) suggested changes in secondary protein structure. Peanut protein–cranberry polyphenol fortified matrices triggered significantly less basophil degranulation than unmodified flour in an ex vivo assay using human blood and less mast cell degranulation when used to orally challenge peanut-allergic mice. Polyphenol fortification of peanut flour resulted in a hypoallergenic matrix with reduced IgE binding and degranulation capacity, likely due to changes in protein secondary structure or masking of epitopes, suggesting potential applications for oral immunotherapy.
Journal information: Journal of Agricultural and Food Chemistry
Provided by American Chemical Society


















