Big results from tiny particles

Creating and manipulating particles made of just a few atoms is all in a day's work for Dr Richard Tilley.
Tilley, an Associate Professor in Victoria's School of Chemical and Physical Sciences, leads the School's nanoparticle and quantum dot research group.
Detecting and treating cancer tumours, finding sustainable energy solutions and creating new products for use in the pharmaceutical and automotive industries are just some of the applications the group is focusing on.
Since his arrival in 2003, Tilley, who is also a principal investigator at the MacDiarmid Institute for Advanced Materials and Nanotechnology, has propelled Victoria's nanoparticle research capability to the forefront of international efforts.
His research group is one of only a few in the world that can create nanoparticles of different sizes and shapes. Its work is supported by large public grants, and its findings are regularly published in the world's top chemistry and nanotechnology research journals.
The group includes Master's, PhD and postdoctoral researchers, collaborators from the MacDiarmid Institute, other New Zealand universities and research institutes, and a number of prestigious international researchers.
The beauty of nanoparticles
Nanoparticles are attracting worldwide scientific interest because of their potential applications, particularly in the fields ofbiomedical research and industrial processes.
"Our goal is to use them to benefit science and, ultimately, mankind," says Tilley.
Nanoparticles' unique properties result from the surface area of their structure. As the size of a material approaches the nanoscale, the percentage of atoms on the surface increases, leading to new chemical and physical properties.
Richard's group uses solution phase chemistry techniques that involve growing small crystals in a liquid solution, then adding what are essentially soap molecules. These bind to the particle surfaces with different strengths. The researchers then carefully manipulate the size and shape, creating thousands of visually striking nanoparticles that react in new ways.
The unique nanoparticle shapes his team is able to create range from squares and pentagons to more elaborate spikey shapes, and possess properties such as increased magnetism and the ability to emit light.
"Even though I have been doing this for more than a decade, I still get a huge buzz every time I look through the microscope at the new particles we can create in the lab," says Tilley.
Detecting tumours through magnetic nanoparticles
One area of success for the group is producing highly magnetic nanoparticles for use in biotechnology applications, such as detecting cancer tissue.
Together with Professor Ian Hermans from the Malaghan Institute of Medical Research, Dr Peter Ferguson at Wellington Public Hospital and researchers from the MacDiarmid Institute and Callaghan Innovation, the team developed a new form of iron metal nanoparticles that don't rust.
The particles can be used as a contrast agent in magnetic resonance imaging (MRI) scans, highlighting the body's internal structures and diagnosing health problems."As a result we have been able to improve performance by 100 percent and detect cancer tumours as small as two millimetres," says Tilley.
"Another benefit is that the magnetic iron nanoparticles can be heated up while in the body, so they have the potential of being used to kill cancer cells, as well as detect them."
One commercial focus for the group is making the catalytic converters in vehicles, which control the toxicity levels of exhaust fumes, more efficient and affordable.
"In a typical converter, only about 10 percent of the nanoparticles are actually active, and they're made from expensive metals such as platinum and palladium," says Tilley. "However, our team has developed a new composite nanoparticle, made from two different elements, that displays much more efficient catalytic properties."
State-of-the-art gadgetry
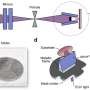
Central to the group's research is Victoria's multimillion-dollar electron microscope suite that houses scanning and transmission electron microscopes that are vital for examining nanomaterials.
The transmission electron microscope, central to the work carried out by Richard's team, allows them to see the size and shape of the particles they create, and to control the properties of the nanoparticles.
The microscope is used to examine ultra-thin samples, and has recently been upgraded.
With higher resolution and a new digital camera, Tilley and his team can now observe nanoparticles at the scale of 0.15 of a nanometre—that is about one billionth of a metre.
"An exciting feature is the camera's faster imaging speed," says Tilley. "That is allowing us to create and analyse videos of nanoparticles moving and interacting for the first time."
The scanning microscopes, used mainly by researchers in the MacDiarmid Institute, have also been improved. These microscopes use a beam of electrons to scan the surface of a sample, gaining information about its shape and composition. A recent addition is a cryoattachment, which rapidly freezes samples, locking their internal structures into solid form and allowing researchers to take images of softmaterials and fluids.
It was previously impossible to look at soft materials, which are important in biological research, because the high vacuum in the microscope caused them to explode.
Looking to the future
Tilley says many students who have worked with the nanoparticles research group have gone on to prestigious postdoctoral positions and successful careers in national and international research institutes and commercial enterprises.
A current focus for the group, says Richard, is commercialisation of its nanoparticles.
Together with PhD graduate Dr Anna Henning, he has launched an online company, Boutiq, to supply nanoparticles for international science and engineering clients. Tilley plans to grow the business through Victoria's commercialisation arm, Vic Link.
"I know how hard it is to actually create something that ends up being used by humanity but, at the same time, I can't imagine ever doing research where I didn't think the outcome would be useful."
Provided by Victoria University