New microbeam emitter has potential to bring promising form of radiation therapy into clinical use
(Phys.org) —Microbeam radiation therapy (MRT) provides tremendous promise for cancer patients through its ability to destroy tumor cells while protecting surrounding healthy tissue. Yet research into its clinical use has been limited by the sheer size of the technology required to generate the beams. Until now, administering MRT required massive electron accelerators known as synchrotrons. But with a new microbeam emitter developed at the University of North Carolina at Chapel Hill, the technology has been scaled down, opening the doors for clinical research.
In a study published online by Applied Physics Letters, a multidisciplinary team led by Physics Professor Otto Zhou, PhD; Radiation Oncology Associate Professor X. Sha Chang, PhD; and Physics Professor Jianping Lu, PhD, built a device using carbon nanotube-based x-ray source array technology developed at UNC that can generate microbeam radiation with similar characteristics as the beams generated by synchrotron radiation. Researchers from Applied Sciences and Radiology at UNC also participated in this study.
Zhou, a member of the UNC Lineberger Comprehensive Cancer Center, points to several studies that have shown that microbeam radiation destroyed tumors and increased survival by as much as a factor of ten in brain tumor bearing animals treated with the technique. While the potential of the technology has been proven, the massive infrastructure needed to undertake the studies has prevented research from being researched for clinical use.
"The innovation here, what we have done at the university, is to build equipment that is compact and can be potentially used in a hospital and achieve similar therapeutic value. The fact that the microbeam can deliver the radiation effect is known, the experiments have been done, but the use of the synchrotron-based equipment is not practical," said Zhou.
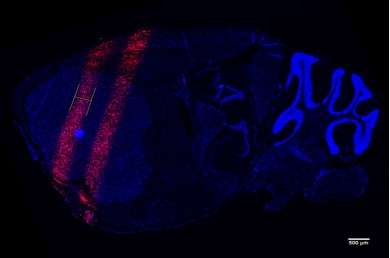
Radiation therapy in current clinical use uniformly bathes tumors in high-dose radiation, but the toxicity from the radiation limits the size of tumors that can be safely treated. MRT doses the area with thin parallel planes of radiation, up to a few hundred microns in width, creating a striped pattern of irradiation across the tumor. In tests with lab animals, this approach leads to lower toxicity and the ability to safely deliver a much higher dose to treat radioresistant tumors such as brain tumors.
The team has already shown that their compact MRT device can generate similar doses and distribution of radiation in lab animals as seen in the synchrotron-based studies, capable of accurately delivering the microbeam radiation to the brain tumor site in mice. With that knowledge, they have begun work on determining whether animals treated with their device show the same therapeutic benefits as those in the synchrotron studies. That would allow researchers at UNC and elsewhere to begin the level of research into MRT necessary to prove that it can benefit human patients.
"Just imagine what could be the potential clinical impact. In many children's brain tumor cancer cases, normally prognosis is not good. Radiation toxicity for developing tissue is much more severe than for adults. This radiation can control the tumor, but it does not damage normal tissue. It has a great potential for clinical application," said Chang, a member of UNC Lineberger.
Because of the limited ability of researchers to study the effects of MRT, little is known about why the radiation destroys tumors but only causes minimal damage to surrounding tissues. A more compact MRT emitter would allow more researchers access to the technology and spur a greater level of research into the interplay between the radiation and biology, according to Lu.
"One of the goals of the research is to understand why it works," said Lu.
Journal information: Applied Physics Letters