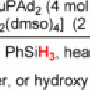Century-old chemistry problem solved

Chemists at The Scripps Research Institute (TSRI) have found a way to apply a "foundational reaction" of organic chemistry to a stubborn class of chemicals, in a transformation that has been thought impossible for a century.
The classic SN2 reaction has enabled chemists to build and modify many pharmaceuticals as well as other useful organic molecules. While the reaction had been thought to exclude certain compounds, a paper in the September 12, 2013 issue of the journal Nature describes a new SN2-like reaction that overcomes this limitation.
"We've widened the range of molecules that are responsive to this foundational technique; for example, we can now chemically synthesize a family of promising antimalarial and anticancer compounds that were previously off limits," said TSRI Assistant Professor Ryan A. Shenvi, who was the senior author of the paper.
Flipping the Umbrella
The SN2 reaction can be used to detach part of a molecule called a functional group from a central carbon atom, while simultaneously, another functional group adds to the opposite side of the carbon atom. This structural flip can significantly change a compound's chemical properties.
"It's like turning an umbrella inside out," Shenvi said. "The SN2 predictably inverts what we call the stereochemistry of the carbon atom."
Traditionally, stereoinversion reactions, which were first described by Paul Walden in 1896, have not been applicable to compounds known as tertiary alcohols or their derivatives—a general problem identified by Christopher Ingold in the early 1900s. In these compounds, the carbon atom of interest is bonded to three other carbon atoms, which effectively shield it from the reaction.
To get around this longstanding limitation, Shenvi and his research associate Sergey V. Pronin developed a method that employs a special acid catalyst plus an unusual nitrogen-containing molecule, a derivative of cyanide. The acid helps detach a fluorous functional group from one side of the central carbon atom, and then the nitrogen forms a new bond on the other side, thus completing the stereoinversion.
"The basic idea is that you can take a tertiary alcohol with one stereochemical configuration and install nitrogen functionality, leaving it with the opposite stereochemical configuration," said Pronin.
Fewer Steps
To demonstrate the new technique, Pronin and graduate student Chris Reiher used it to build or modify a number of compounds in a relatively short sequence of reaction steps, starting with cheaply available tertiary alcohols. "In one example, we took a derivative of vitamin E, tocopherol, and in a few steps turned it into something we call an aza-tocopherol, in which the oxygen atom is replaced with nitrogen—a compound that otherwise would have been very difficult to access," Pronin said.
The chemists also showed how the new reaction can simplify the preparation of a set of compounds known as marine isocyanoterpenes, which are produced naturally by sea sponges and other oceanic animals. Some of these compounds have been found to have anticancer, antimalarial, antifungal and other potentially useful properties, but they have been very hard to prepare using synthetic chemistry. With the new technique, the researchers were able to prepare several scarce marine isocyanoterpenes starting from abundant and renewable terrestrial terpenes, and using far fewer steps than had ever been reported before.
On its own, the new method fills a significant gap in the toolkit of organic chemists. But Shenvi hopes soon to extend it further to enable SN2-like substitutions of tertiary alcohols with other reaction partners: "We think that this reaction will teach us how to achieve stereoinversions of tertiary carbons to form carbon-oxygen, carbon-sulfur and even carbon-carbon bonds," he said.
This year would have been Paul Walden's 150th birthday.
More information: Paper: Stereoinversion of tertiary alcohols to tertiary-alkyl isonitriles and amines, DOI: 10.1038/nature12472
Journal information: Nature
Provided by The Scripps Research Institute