New inexpensive, efficient catalysts offer viable way to store and reuse renewable energy

Two University of Calgary researchers have developed a ground-breaking way to make new affordable and efficient catalysts for converting electricity into chemical energy.
Their technology opens the door to homeowners and energy companies being able to easily store and reuse solar and wind power. Such energy is clean and renewable, but it's available only when the sun is shining or the wind is blowing.
The research by Curtis Berlinguette and Simon Trudel, both in the chemistry department in the Faculty of Science, has just been published in Science.
"This breakthrough offers a relatively cheaper method of storing and reusing electricity produced by wind turbines and solar panels," says Curtis Berlinguette, associate professor of chemistry and Canada Research Chair in Energy Conversion.
"Our work represents a critical step for realizing a large-scale, clean energy economy," adds Berlinguette, who's also director of the university's Centre for Advanced Solar Materials.
Simon Trudel, assistant professor of chemistry, says their work "opens up a whole new field of how to make catalytic materials. We now have a large new arena for discovery."
The pair have patented their technology and created from their university research a spin-off company, FireWater Fuel Corp., to commercialize their electrocatalysts for use in electrolyzers.
Electrolyzer devices use catalysts to drive a chemical reaction that converts electricity into chemical energy by splitting water into hydrogen and oxygen fuels. These fuels can then be stored and re-converted to electricity for use whenever wanted.
The only byproduct from such a 'green' energy system is water, which can be recycled through the system.
To store and provide renewable power to a typical house would require an electrolyzer about the size of a beer fridge, containing a few litres of water and converting hydrogen to electricity with virtually no emissions, the researchers say.
Key to their discovery is that they deviated from conventional thinking about catalysts, which typically are made from rare, expensive and toxic metals in a crystalline structure.
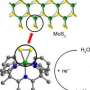
Instead, Berlinguette and Trudel turned to simpler production methods for catalysts. This involved using abundant metal compounds or oxides (including iron oxide or 'rust'), to create mixed metal oxide catalysts having a disordered, or amorphous, structure.
Laboratory tests – reported in their Science paper – show their new catalysts perform as well or better than expensive catalysts now on the market, yet theirs cost 1,000 times less.
Their research was supported by the university's Institute for Sustainable Energy, Environment and Economy, Alberta Innovates, Mitacs and FireWater Fuel Corp.
FireWater Fuel Corp. expects to have a commercial product in the current large-scale electrolyzer market in 2014, and a prototype electrolyzer – using their new catalysts – ready by 2015 for testing in a home.
Research details:
What have the two University of Calgary researchers discovered?
They have discovered a ground-breaking way to make new affordable and highly efficient catalysts (called electrocatalysts) for converting electricity into chemical energy. A catalyst is a substance that increases the rate of a chemical reaction.
Why are electrocatalysts useful?
Electrocatalysts are used in electrolyzers, devices that split water into hydrogen and oxygen through a chemical reaction driven by electricity. The hydrogen can then be stored and re-converted to electricity for use whenever wanted.
Scientists have been working for several decades on the problem of trying to make efficient and inexpensive electrocatalysts.
Today's commercial electrocatalysts are typically made of crystalline metal oxides (any chemical compound that has a metal) using rare, expensive and/or toxic metals (e.g. ruthenium, iridium). Such catalysts work well but their prohibitive cost makes them impractical for widespread use, such as in homes and by businesses.
What makes the electrocatalysts created by the U of Calgary researchers different than conventionally made, commercial catalytic materials?
Chemists have traditionally been attracted to creating catalysts out of 'pure' crystalline-structured materials. They've tended to ignore unstructured material as the "crud at the bottom of the flask."
"There really have been few significant advances in catalyst design over the last three decades," Berlinguette says.
He and Trudel developed a novel process that uses cheap, abundant and non-toxic metals (e.g. iron, cobalt, nickel) combined in a highly disordered, or amorphous, structure.
Think of crystalline structures as being like tiles laid in an ordered pattern on a floor, while amorphous structures are like tiles thrown on a floor. Such an amorphous material has no symmetry and is full of 'defects.'
These 'defects' in amorphous mixed metal oxide materials actually make them more chemically reactive – and therefore more efficient catalysts – than crystalline materials.
Laboratory tests by the U of Calgary researchers show their catalysts perform as well as or better than catalysts now on the market – but theirs are 1,000 times cheaper.
"We're essentially showing, even with our 'first generation' of catalysts, that we're equal to or better than anything that's sold commercially right now after 30 years of development," Trudel says.
The researchers say they can utilize any metal in the periodic table and are able to combine as many metals as they want into their catalysts.
"Our fabrication method provides access to an entirely new domain of catalytic materials," Berlinguette says.
What is the significance of this discovery?
The U of Calgary researchers are the first group in the world to utilize their scalable photochemical process to make heterogeneous mix-metal amorphous electrocatalysts for clean hydrogen production.
"As far as we know, there is no other method to easily make amorphous materials where we can combine the metals in any ratio we desire. Now the 'fun' is trying to hit the composition that produces the best catalysts," Trudel says.
What application does their discovery have in the 'real' world?
Having cheap and efficient electrocatalysts would enable homeowners and energy companies to store and reuse, whenever needed, intermittently generated electricity such as solar and wind power.
There is currently no inexpensive way of storing such renewable energy. So electricity generated by the sun or the wind is available only when the sun is shining or the wind is blowing.
Electrocatalysts are used in devices called electrolyzers, which convert electricity into chemical energy by splitting water into hydrogen and oxygen fuels. These fuels can then be stored and reconverted to electricity for use whenever wanted. The only byproduct of such a 'green' energy system is water, which can be recycled through the system.
"This is a completely repeatable and carbon-neutral cycle . . . it's not using carbon at all," Trudel says.
Batteries can also be used to store electricity generated by wind and solar power. However, current battery technology is very inefficient compared with hydrogen, which can store much more energy than batteries.
"The principal role of hydrogen in the energy economy is the storage of solar and wind energy," Berlinguette says.
Cheap and efficient catalysts would provide homeowners and businesses with affordable electrolyzers.
"People could actually start storing renewable energy when it's available and keep that in their house all day and take advantage of it at night," Trudel says.
For example, all of Alberta's wind power farms are located in the southern region of the province where wind conditions are optimum. But when the wind blows, they all produce electricity at the same time.
That drives down the price at which wind farm operators can sell their power to the provincial electrical grid.
What if operators had an affordable way to store that wind-generated electricity, using an electrolyser?
They could then store their wind power as hydrogen, and reconvert to electricity when there's greater demand and they can get a higher price for their clean power.
Electrolyzers with cheap, efficient catalysts could be sized to a homeowner's furnace room, or scaled up to a tractor trailer-sized unit that would store renewable power as hydrogen for reuse by a community, in a 'green' district energy system.
"Electrolyzers effectively enable you to purchase electricity at a discounted rate when there is no demand, and sell back to the grid at peak times," Berlinguette says.
What are the next steps for the U of Calgary researchers?
The researchers are testing various formulations of their amorphous mixed metal oxide catalysts, to better understand the materials and design the optimal catalysts. This includes using proven nanotechnology methods to increase the amount of hydrogen produced.
They also are working toward making a "photo-electrocatalyst," which uses sunlight to increase the hydrogen produced by the electrolyzer.
They have patented both their process for creating their electrocatalysts as well as the new catalysts they've created, and they've established a spin-off company, FireWater Fuel Corp., to commercialize their technology.
The company expects to have a commercial product in the current large-scale electrolyzer market in 2014, and a prototype electrolyzer – using their new catalysts – ready by 2015 for testing in a home.
More information: "Photochemical Route for Accessing Amorphous Metal Oxide Materials for Water Oxidation Catalysis," by R.D.L. Smith et al, Science, 2013.
Journal information: Science
Provided by University of Calgary