Using the world's rarest element to study the world's rarest force
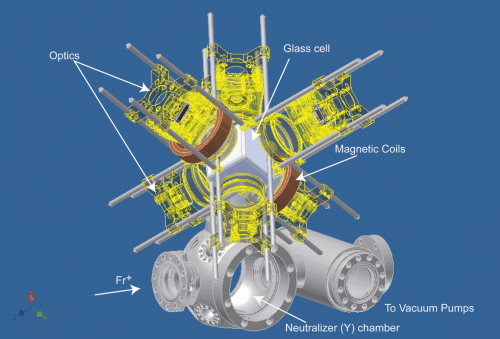
In September 2012 an experiment at the TRIUMF accelerator complex reached a milestone. The FrPNC collaboration —-short hand for Francium Parity Non-conservation—-succeeded in trapping several kinds of francium, including the isotope Fr-207, which had never before been trapped. There is less than a gram of Fr at any given time in the whole earth. With a halflife of mere seconds, and existing only briefly at particle accelerators, francium is the rarest species in the periodic table of elements up to uranium.
Luis Orozco helped to pioneer the close study of francium back in the 1990s. A JQI fellow and professor at the University of Maryland, Orozco played a large role in the design of the present experiment and leads the JQI contingent now at work in Vancouver.
So what does francium have to offer that makes it worth the trouble of dealing with so ephemeral a substance? It turns out that Fr, element 87 and the heaviest of all the alkalis, is the best type of atom for studying the breakdown of parity, a fundamental property of nature, and for studying the weak nuclear force responsible for the breakdown.
Transmutation
Because francium falls apart so quickly it has be to be made fresh over and over, like baguettes. In this regard TRIUMF acts as a foundry, transmuting not lead into gold but uranium into francium. First a beam of protons with an energy of 500 million electron volts (500 MeV) is smashed into a target made of uranium and carbon. The energy of impact jars loose some constituent particles from the uranium atoms, turning some of them into dozens of other atomic species, including several isotopes of francium, including Fr-207, Fr-209, and Fr-221 (the numbers giving the total number of protons and neutrons in the atomic nucleus), which can be sorted and selected using magnetic fields.
Thereafter the process of harvesting and studying francium is, in effect, an elaborate act of cooling. At TRIUMF about 100 million Fr atoms are made per second. These are gathered together into a beam with an energy of about 30 keV. These charged atoms (ions) enter a neutralization chamber where they strike a tiny sample of the element yttrium. Here the Fr ions become neutral atoms, cool, and then percolate to the surface of the Y sample, where they emerge as a warm gas at a temperature of 700 C.
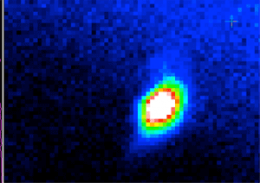
This temperature, equivalent to an energy of a fraction (about 1/10th of an eV), is still too high for the Fr atoms to be studied. A set of lasers cool the atoms further by bombarding them gently, slowing their motions while they are held in place in conjunction with a series of magnetic fields. This whole apparatus is thus referred to as a magneto-optic trap (MOT). The picture here shows the fluorescence a blob of hundreds of thousands of Fr atoms as they are held in the trap.
Parity Violation
Cooled now to a temperature of only 10 milli-kelvin, Fr atoms can serve as a tiny laboratory for studying the weak nuclear force. The four known physical forces can be depicted as carried from place to place by special particles: (1) the electromagnetic force is carried by photons (the particle manifestation of light); the strong nuclear force is carried by gluons; the weak nuclear force is carried by a fleet of three heavy particles called W+, W-, and Z0; and gravity is carried by gravitons, which have not yet been detected.
Besides their very different magnitudes, these forces differ in some of their other effects. Parity—-the proposition that nature cannot tell left from right—-is upheld by the strong and electromagnetic forces but not by the weak force. That is, for parity to be "conserved," then it would make no difference in our measurements whether we were observing an interaction among particles directly or in a mirror. For the strong and electromagnetic forces this is true but not for the weak force. The weak force, although not weaker than gravity, is the least palpable of the forces in our ordinary experience. It operates only within the nuclei inside atoms and is therefore hard to probe. Consequently physicists welcome the chance for any purchase they can get on this elusive force. For many years parity violation was observed only in the decay of certain nuclei. But it is also desirable to study parity-non-conservation in atoms. Many believe that because of the underlying nature of francium, it is the best atom in which to study such parity effects.
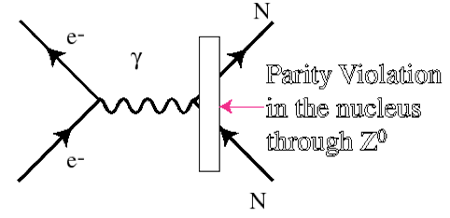
Feynman diagrams are a handy way of depicting forces at work. In this drawing an electron inside an atom is depicted as interacting with the nucleus (or with a single part of a nucleus such as a proton or neutron) designated by N. The Greek gamma stands for a photon. But insofar as the electromagnetic and weak forces are considered as being two aspects of one combined "electroweak" force, the electron can interact with the nucleus, at least part of the time, via the (parity-conserving) electromagnetic force and part of the time via the (parity-non-conserving) weak force. Still another way of saying this is that the electron can interact with constituent quarks (letter q) within a proton via the exchange of a Z particle, as depicted here.
Studying this fundamental interaction is what the FrPNC experiment is doing at TRIUMF. In effect it undertakes at very low energies (nano-electron-volts) what normally happens at very high energy (billions of electron volts), a branch of science called high-energy physics.
Provided by University of Maryland



















