How species evolve: Epilogue to 'On the Origin of Species'?
(PhysOrg.com) -- One of the grand challenges in biology is to understand how species evolve. Today, more than 150 years after the publication of Darwin’s ‘On the Origin of Species’, we still do not truly understand the speciation process.
This stems partly from the fact that most classical studies of speciation are based on species that have already diverged leaving scientists little choice but to speculate back in time to infer the causes of speciation.
Conway Fellow, Dr Emma Teeling has led a project, funded by an SFI Research Frontiers Project grant, with international collaborators in Thailand, Myanmar, France and the United Kingdom to address this fundamental question in biology with global conservation implications.
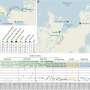
The findings of their unique study that captures speciation ‘in action’ in ecologically diverging populations of the world’s smallest mammal, the bumble-bee bat (Craseonycteris thonglongyai) in Thailand and Myanmar were published recently in Nature Communications.
These populations represent a ‘one-of-its-kind’ natural experiment in terms of ‘capturing’ the appropriate evolutionary timescale to enable us to identify the evolutionary processes that drive speciation in nature.” says Dr. Emma Teeling.
By investigating the early drivers of speciation at different evolutionary time-scales, the study shows that limited gene flow, resulting from geographic distance, is initially required to promote sensory ecological speciation.
“To do this, we examined the spatial structure, genetic architecture and sensory ecological traits between and within the only two known populations of the world’s smallest mammal, the bumble-bee bat. We generated and collected a weight of molecular, ecological and acoustic data and show that geographic distance has a primary role in limiting gene flow rather than echolocation divergence.
Our results support sensory ecology acting as a reinforcement-like mechanism in the speciation process rather than being the main driver as was previously hypothesized in other well-documented empirical studies. Our results question whether true sympatric speciation actually occurs or if some level of geographic isolation and thus restricted gene flow is always necessary to initiate the speciation process” says Dr. Sebastien Puechmaille, the first author on this study.
The study also identified for the first time a candidate ‘echolocation’ gene (RBP-J) that shows evidence of divergent selection matching the echolocation divergence within the Thai population.
This is the first reported association of this gene with echolocation capabilities. This gene is involved in hair cell formation in the cochlea (sound receiving organ in the inner-ear). As bats are using the highest frequencies (above 200 kHz) of all mammals, their hearing system, particularly the hair cells in the organ of Corti where the sound is received and amplified, needs to be well adapted.
“We also show that inter-specific competition with another bat species, Myotis siligorensis drives the local sensory adaptation as opposed to random drift or abiotic factors such as temperature and humidity,” says Dr. Sebastien Puechmaille.
From a conservation perspective, this study is the first to investigate the population structure and evolutionary history of this tiny mammal.
“It is a rare and endangered, charismatic bat species, restricted to a 2000 km2 region that saddles the Thai-Myanmar border and is considered as one of the top ten Evolutionary Distinct and Globally Endangered (EDGE) species,” adds Dr. Emma Teeling.
Evolutionary analyses of maternal, paternal, biparentally inherited genetic markers and ecological data support the presence of two species of bumble-bee bat, one in Thailand and one in Myanmar, which split approximately 0.4 million years ago. Limited dispersal capabilities and restricted ranges (less than 2000 km2) suggest that both species are under threat and warrant separate conservation management plans.
More information: Puechmaille et al. The evolution of sensory divergence in the context of limited gene flow in the bumblebee bat Nature Communications (2011) doi:10.1038/ncomms1582
Journal information: Nature Communications
Provided by University College Dublin