New form of hafnium oxide developed

(PhysOrg.com) -- A novel material developed by researchers at the University of Cambridge is opening up new possibilities for next generation electronic and optoelectronic devices, and paving the way for further component miniaturisation.
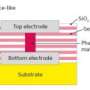
The material, a new form of hafnium oxide, was developed by Dr Andrew Flewitt’s research group in the Department of Engineering. The material provides exceptionally high dielectric constant compared with currently existing forms of hafnium oxide, which is already a key material in the electronics industry.
Metal oxides are used in a wide variety of applications. Normally, they are produced on substrates by sputtering, a process by which some of the atoms of an electrode are ejected as a result of bombardment by heavy positive ions. One of the problems when attempting to make high-quality electronic materials through sputtering, however, is the difficulty in precisely controlling the energetics of the deposition process, and hence the material properties such as defect density.
In order to enable much greater control of the material properties, Dr Flewitt and his team began using a novel deposition technology to promote plasma sputtering. The technology, known as HiTUS (High Target Utilisation Sputtering), was developed by a UK-based company, Plasma Quest Ltd. One of the first materials that the Cambridge team looked at using HiTUS was hafnium oxide.
Hafnium oxide is an electrical insulator which is used in optical coatings, capacitors and transistors, among other applications. Many companies are currently using hafnium oxide to replace silicon dioxide in transistors, due to its high ratio of electric displacement in a medium to the intensity of the electric field producing it, known as a dielectric constant. The higher the dielectric constant of a material, the higher its capacitance - the ability to store an electric charge.
Hafnium oxide forms in different crystalline and polycrystalline structures: monoclinic, cubic and orthorhombic. However, an amorphous form is preferable to polycrystalline forms due to the absence of grain boundaries, the point at which two crystals in a polycrystalline material meet. Grain boundaries act as conduction paths through thin films of the material. They not only reduce the resistivity, but lead to a non-uniformity in conductivity over a large area, which itself leads to spatial non-uniformity in device performance However, until now amorphous hafnium oxide has had a relatively low dielectric constant of around 20.
The form of hafnium oxide developed by Dr Flewitt has a dielectric constant higher than 30.
“Most people thought that all amorphous hafnium oxide had to exist in the monoclinic-like phase,” says Dr Flewitt. “What we’ve shown is that it can exist and does exist in a cubic-like phase. This is similar to amorphous carbon, where you can get diamond-like properties out of amorphous carbon material.”
Amorphous dielectrics are more homogenous than other forms, allowing improved uniformity from one device to another, and the absence of grain boundaries results in higher effective resistivity, as well as less optical scatter.
The material is produced using a room-temperature, high-deposition rate process, making it particularly suitable for plastic electronics and high-volume semiconductor manufacturing. The absence of grain boundaries makes the material ideal for optical coatings and more efficient solar cells.
Cambridge Enterprise, the University’s commercialisation group, is currently seeking commercial partners for collaborative development and licensing of this material.
Provided by Cambridge University