New evidence that bacteria in large intestine have a role in obesity
Bacteria living in people's large intestine may slow down the activity of the "good" kind of fat tissue, a special fat that quickly burns calories and may help prevent obesity, scientists are reporting in a new study. The discovery, published in ACS' Journal of Proteome Research, could shed light on ways to prevent obesity and promote weight loss, including possible microbial and pharmaceutical approaches, the authors said.
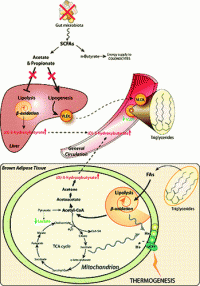
Sandrine P. Claus, Jeremy K. Nicholson and colleagues explain that trillions of bacteria live in the large intestine of healthy people, where they help digest food and make certain vitamins. In recent years, however, scientists have realized that these bacteria do more — they interact with the rest of the body in ways that affect the use of energy and its storage as fat and finely tune the immune system. Claus and Nicholson decided to see how intestinal bacteria might affect the activity of brown fat. The "good" fat that burns calories quickly before they can be stored as fat, brown fat exists in small deposits in the neck area and elsewhere — not like "white fat" in flab around the waist and buttocks. No one had checked to see if those bacteria could have an effect on brown fat, the researchers noted.
In experiments that compared "germ-free" (GF) mice, which don't have large-intestine bacteria, and regular mice, the scientists uncovered evidence suggesting that the bacteria do influence the activity of brown fat. Brown fat in the GF mice seemed to be more active, burning calories faster than in regular mice. Large-intestine bacteria also seemed to be linked with gender differences in weight. Normal male mice were heavier and fattier than females, but those differences vanished in the GF mice. The research also uncovered major differences in the interactions between males and females and their intestinal bacteria that might help explain why the obesity epidemic is more serious and rapidly developing in women. Those and other findings may point the way toward approaches that kick-up the activity of brown fat in humans to prevent or treat obesity.
More information: Gut Microbiota Modulate the Metabolism of Brown Adipose Tissue in Mice, J. Proteome Res., Article ASAP. DOI: 10.1021/pr200938v
Abstract
A two by two experimental study has been designed to determine the effect of gut microbiota on energy metabolism in mouse models. The metabolic phenotype of germ-free (GF, n = 20) and conventional (n = 20) mice was characterized using a NMR spectroscopy-based metabolic profiling approach, with a focus on sexual dimorphism (20 males, 20 females) and energy metabolism in urine, plasma, liver, and brown adipose tissue (BAT). Physiological data of age-matched GF and conventional mice showed that male animals had a higher weight than females in both groups. In addition, conventional males had a significantly higher total body fat content (TBFC) compared to conventional females, whereas this sexual dimorphism disappeared in GF animals (i.e., male GF mice had a TBFC similar to those of conventional and GF females). Profiling of BAT hydrophilic extracts revealed that sexual dimorphism in normal mice was absent in GF animals, which also displayed lower BAT lactate levels and higher levels of (D)-3-hydroxybutyrate in liver, plasma, and BAT, together with lower circulating levels of VLDL. These data indicate that the gut microbiota modulate the lipid metabolism in BAT, as the absence of gut microbiota stimulated both hepatic and BAT lipolysis while inhibiting lipogenesis. We also demonstrated that 1H NMR metabolic profiles of BAT were excellent predictors of BW and TBFC, indicating the potential of BAT to fight against obesity.
Journal information: Journal of Proteome Research
Provided by American Chemical Society
















