How algae use a 'sulfate trap' to selectively biomineralize strontium
(PhysOrg.com) -- In any kind of nuclear reactor, there is a small amount of the radioactive isotope strontium-90 that is formed as part of the regular fission process. In fact, fission products such as strontium-90 make up approximately 3% of the total mass of spent nuclear fuels. The majority of the fission products are stable isotopes or very short-lived radioisotopes that do not cause much concern. However, there are several radioactive fission products that are present in significant quantities in spent fuels, including strontium-90 and caesium-137 (which together make up 0.3% of the total mass), as well as iodine-129 and technetium-99 (which together make up 0.1% of the total mass).
Strontium-90 has a half-life of approximately 30 years, and emits a beta particle as it decays to yttrium-90 (another radioisotope). It is particularly hazardous because in our bodies, strontium is treated the same way as calcium. In fact, the chemistry of strontium is so similar to calcium that it is very difficult to separate the two, even in a lab. If ingested, strontium can become stored in bones, where it is very stable, and may remain there for the rest of one’s life. While some isotopes of strontium are harmless, strontium-90 poses serious health risks such as cancer, and may continue to do so years after the initial exposure.
Over the years, researchers have looked to a small number of organisms, including the desmid green alga Closterium moniliferum, because of their ability to selectively sequester strontium. Somehow, these algae were able to take up and immobilize strontium from samples containing both calcium and strontium, a skill that could potentially be used to reduce the volume of dangerous strontium waste. However, it was not clear how the algae were capable of doing this.
Now, researchers at Northwestern University and Argonne National Laboratory have uncovered the secrets of the algae using x-ray fluorescence microscopy (XFM) at the U.S. Department of Energy Office of Science’s Advanced Photon Source (APS) at Argonne. This work, which was recently featured on the cover of the Journal of Structural Biology, utilized the x-ray fluorescence microprobe at X-ray Science Division (XSD) beamline 2-ID-E, and the sulfur K-edge x-ray absorption near-edge spectroscopy (XANES) capability of the scanning microprobe at XSD beamline 2-ID-B.
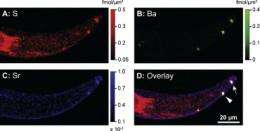
Using XFM, the researchers were able to quantitatively measure the amounts of strontium and sulfur at every point in the cell, creating a “map” of these elements in the cell. They were able to correlate this with electron microscopy images, and determine the concentrations of strontium and sulfur in the vacuole, or storage compartment of the algae. They also used sulfur-XANES to determine that the sulfur in the vacuole was inorganic sulfate, rather than part of a protein. By performing measurements both under conditions where the strontium was biomineralized by the cell into crystals, and where it was not, they were able to determine that it was actually the sulfate concentration in the vacuole that was causing selective sequestration of the strontium. Thus, the alga was selectively isolating the strontium into crystals by controlling the amount of sulfate in its vacuole, rather than utilizing any other kind of strontium-specific transport mechanism. This kind of selectivity is called a “sulfate-trap” mechanism.
Now that the mechanism is understood, the researchers postulate that the algae could be used to remediate contaminated environments. Requiring only light and water to carry out their task, these algae would be an energy-efficient solution to a environmental problem, but further work, such as determining the algae’s hardiness to radiation remains to be done before it is clear whether these applications are practical.
More information: Minna R. Krejci, Brian Wasserman, Lydia Finney, Ian McNulty, Daniel Legnini, Stefan Vogt, Derk Joester, “Selectivity in biomineralization of barium and strontium,” J. Struct. Biol. 176, 192 (2011). DOI:10.1016/j.jsb.2011.08.006
Provided by Argonne National Laboratory


















