Adding neutrons to synthetic atoms drastically alters shape of their nuclei, affects their stability
To probe the evolution of atomic nuclei with different shape -- a factor which affects atomic stability -- a large team of international researchers has added neutrons to zirconium atoms and revealed the possibility of very unusual shapes. "The shape of a nucleus reflects the symmetry of its quantum state," explains team member Hiroyoshi Sakurai from the RIKEN Nishina Center for Accelerator-Based Science in Wako, Japan. This result helps us to understand how many neutrons are needed for the most stable nuclei.
Most atoms can exist in one of several alternative forms called isotopes, depending on the number of neutrons in their core. Naturally occurring, stable, atoms tend to have between 1 and 1.5 neutrons per proton. However, synthetically generated atoms with higher neutron–proton ratios can reveal much about changes within an atomic nucleus.
The protons and neutrons in a nucleus usually form arrangements of concentric spherical shells. In some cases, however, the outermost particles exist further from the center than normal. This can lead to nuclei that are wider than they are long. Just as atoms with a specific number of protons can exist as different isotopes, atoms with a specific number of protons and neutrons can exist as different nuclear isomers—nuclei with different shapes. “By measuring the shape of nuclei, we are probing the internal symmetry in the nucleus—the so-called shell structure,” explains Sakurai.
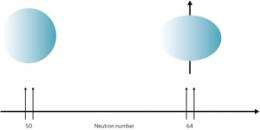
At the Radioactive Isotope Beam Factory in Japan, operated jointly by RIKEN and The University of Tokyo, the researchers experimented with zirconium atoms that have 40 protons and, in their stable form, between 50 and 52 neutrons. They created zirconium atoms with as many as 68 neutrons through collisions between uranium and beryllium atoms. After filtering isotopes from the remnants of the collision, they measured the rate of decay of beta and gamma radiation emitted by the quickly decaying, unstable synthetic atoms. The measurements showed that these nuclei changed shape from spherical to oblate.
The degree of deformation of the zirconium nuclei increased as Sakurai and colleagues added more neutrons, but this trend stopped when they reached 64 neutrons. This result raises the intriguing prospect of a tetrahedral-shaped isomer of zirconium-108—an isotope with 68 neutrons—which has been predicted previously by other researchers. However, further work is needed to verify this.
“We next hope to gain further insight into the evolution of nuclear isomers by extending our study to strontium atoms,” Sakurai says.
More information: Sumikama, T., Yoshinaga, K., Watanabe, H., Nishimura, S., Miyashita, Y., Yamaguchi, K., Sugimoto, K., Chiba, J., Li, Z., Baba, H. et al. Structural evolution in the neutron-rich nuclei 106Zr and 108Zr. Physical Review Letters 106, 202501 (2011). prl.aps.org/abstract/PRL/v106/i20/e202501
Provided by RIKEN