July 13, 2011 feature
Rechargeable lithium-sulfur batteries get a boost from graphene
(PhysOrg.com) -- By wrapping tiny sulfur particles in graphene sheets, researchers from Stanford University have synthesized a promising cathode material for rechargeable lithium-sulfur batteries that could be used for powering electric vehicles on a large scale. When combined with silicon-based anodes, the new graphene-sulfur cathodes could lead to rechargeable batteries with a significantly higher energy density than is currently possible.
The researchers, led by Yi Cui and Hongjie Dai from Stanford University, have published their study in a recent issue of Nano Letters.
As the researchers explain in their study, in order to power electric vehicles that are competitive with gasoline-powered vehicles, one of the biggest challenges is improving the energy and power densities of rechargeable lithium batteries. The batteries’ weak spot is currently the cathode materials, which have specific capacities that are much lower than those of the anode materials. (The specific capacities for cathode materials are about 150 mAh/g for layer oxides and 170 mAh/g for LiFe-PO4, while those for anode materials are 370 mAh/g for graphite and 4200 mAh/g for silicon.)
In order to improve the cathode, the researchers turned to sulfur, which has a theoretical specific capacity of 1672 mAh/g, about five times higher than those of traditional cathode materials. Although sulfur has other advantages, such as low cost and a benign environmental impact, it also has some disadvantages. For instance, sulfur is a poor conductor, it expands during discharge, and the polysulfides dissolve in electrolyte. Together, these problems cause a low cycle life, low specific capacity, and low energy efficiency.
Previous research has shown that adding carbon to sulfur can increase sulfur’s electrical conductivity. But although various carbon-sulfur composites have achieved specific capacities of more than 1000 mAh/g, their cycle life is still low; it remains challenging to retain these high capacities for more than 100 cycles.
“We developed a strategy of graphene wrapping to overcome many issues related to using sulfur as lithium-ion battery cathodes,” Cui told PhysOrg.com. “We have shown excellent cycling performance.”
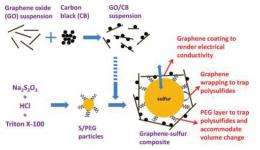
To achieve this high performance, the Stanford researchers made some adjustments to the sulfur. First, they coated submicrometer sulfur particles with poly(ethylene glycol) (PEG) to trap the polysulfides and prevent their dissolution. The flexible PEG coating also improves cycle life by accommodating the sulfur particles’ volume expansion during the discharge portion of each cycle. Next, the researchers wrapped the coated sulfur particles with graphene sheets decorated with carbon black nanoparticles, which improves the sulfur cathode’s conductivity. The loosely packed graphene layer also further traps polysulfides and accommodates the volume expansion of the sulfur.
“This is a very rational material design to overcome the issues of polysulfide dissolution,” said Hailiang Wang, lead author of the paper.
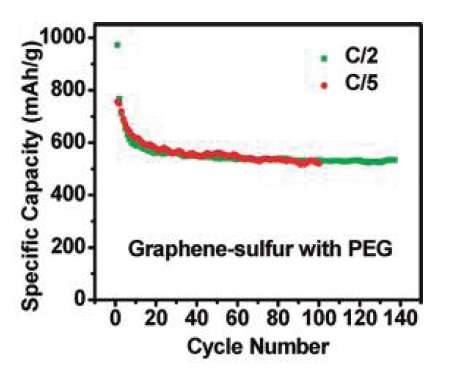
The researchers demonstrated that the resulting graphene-sulfur cathode can achieve high specific capacities of 500- 600 mAh/g for more than 100 cycles. The new cathode material could be used to fabricate rechargeable batteries with a higher energy density than that of other rechargeable batteries today.
“The capacity fading is only about 10-15% for 100 cycles, which is very exciting,” said coauthor Yuan Yang, who made electrodes and cells in the project.
However, before such batteries can be manufactured, the researchers have to address the large performance variability of the lithium-sulfur batteries that they tested in this study. For example, about 30-50% of the batteries had a 20-25% decay over 100 cycles. In the future, the researchers hope to continue improving the capping of sulfur to enable lossless cycling.
“Overall, the biggest challenges facing rechargeable batteries for electric vehicles are increasing the energy density and reducing the cost,” Cui said. “Using high-energy and low-cost materials such as sulfur is very attractive.”
More information: Hailiang Wang, et al. “Graphene-Wrapped Sulfur Particles as a Rechargeable Lithium-Sulfur Battery Cathode Material with High Capacity and Cycling Stability.” Nano Letters, DOI: 10.1021/nl200658a
Copyright 2011 PhysOrg.com.
All rights reserved. This material may not be published, broadcast, rewritten or redistributed in whole or part without the express written permission of PhysOrg.com.