Organic chemistry: Amino acids made easy
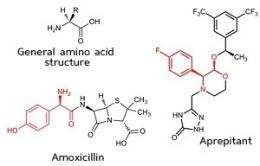
Amino acids are the building blocks of proteins. There are 22 different amino acids and they can combine in a myriad ways to form a vast array of proteins. All amino acids except glycine are chiral molecules, meaning they exist in two mirror-image, or enantiomeric, forms—only one of which is naturally occurring. These unnatural enantiomers of amino acids are in great demand by the pharmaceutical industry as the raw materials for the production of a variety of drugs, including the antibiotic amoxycillin and the anti-nausea drug aprepitant (see image).
One method that is widely used to produce amino acids generally is Strecker synthesis—a chemical reaction devised by the nineteenth-century German chemist Adolph Strecker that combines an aldehyde, ammonia and hydrogen cyanide to produce an aminonitrile that can be easily converted into an amino acid. Unfortunately, the original Strecker synthesis can only produce a mixture of the enantiomeric forms of an amino acid. For this reason, many chemists have taken an interest in the development of enantioselective, or asymmetric, catalytic reactions—reactions that use a catalyst to selectively increase the formation of a particular enantiomer.
Some catalytic enantioselective variations of Strecker synthesis have already been reported, but there are problems. Many require the use of expensive sources of cyanide—typically a compound called trimethylsilylcyanide—and very low temperatures, which can be difficult to achieve on an industrial scale.
Abdul Majeed Seayad at the A*STAR Institute of Chemical and Engineering Sciences and co-workers have now developed an asymmetric Strecker protocol that uses hydrogen cyanide as the cyanide source and which proceeds at room temperature. The new methodology still requires the use of trimethylsilylcyanide, which the researchers found to be essential to achieving an enantioselective reaction, but only a relatively small catalytic amount is required and it is regenerated in the reaction by the addition of cheaper hydrogen cyanide. Seayad and his co-workers showed that they can use their conditions to produce a variety of unnatural amino acids.
As with most methodology developments, there is room for improvement with further research. “So far we’ve tackled only amino acids with aromatic side-chains,” explains Seayad. “We would like to develop the process to produce amino acids with other side chains. Hydrogen cyanide is inexpensive but it is extremely toxic and special equipment and training are needed to handle it. We are exploring ways in which we might generate it in the reaction, which would be much safer.”
More information: Ramalingam, B. et al. A remarkable titanium-catalyzed asymmetric Strecker reaction using hydrogen cyanide at room temperature. Advanced Synthesis and Catalysis 352, 2153–2158 (2010). dx.doi.org/10.1002/adsc.201000462
Provided by Agency for Science, Technology and Research (A*STAR)



















