Accessing the risk of arsenic ingestion with mineralogy
Canadian researchers working at Brookhaven's National Synchrotron Light Source (NSLS) have created a method for determining how much of the arsenic in soil tailings -- byproducts of the mining industry -- will enter the bloodstream if ingested.
"People tend to get frightened when they hear about chemicals in the environment, but those chemicals have to get in the bloodstream first," explained lead researcher Kenneth Reimer, director of the Environmental Sciences Group at the Royal Military College of Canada. "The default assumption is that all arsenic attached to ingested particles will enter your system, but you're going to eliminate some of it through waste."
Those who live near past and present mining sites may accidentally consume soil samples containing arsenic. (In fact, children eat an average of 80 milligrams of dirt a day, four times as much as adults.) In order to ensure that harmful mining waste is properly dealt with, it is important to determine how much of an ingested chemical is absorbed by the body rather than passed harmlessly through the digestive system. Scientists can approximate this as the chemical's bioaccessibility.
"Bioaccessibility gives us a much better idea of the actual risk involved in arsenic ingestion," explained Reimer. "It's much more accurate than just using the default assumption of 100 percent absorption."
Bioaccessibility testing approximates the percentage of an ingested chemical that will enter the body, without requiring animal or human testing. Instead of feeding samples to a test subject, a bioaccessibility test runs the sample through conditions that simulate a digestive system, complete with mock gastrointestinal fluids. The percentage of arsenic that dissolves during this process reveals the sample's bioaccessibility.
But what if, instead of running these tests on numerous soil samples from throughout a mining site, researchers could describe the relationship between a sample's bioaccessibility and the specific minerals in it — its mineralogy? Reimer and his team sought to do so, examining the bioaccessibility and mineralogy of soil samples taken in the gold mine districts of Nova Scotia, Canada.
Simply put, Reimer and his team used this study to ask, "Could we predict, given the mineralogy of a sample, whether it's highly bioaccessible?"
The research team tested the samples' mineralogy using micro-x-ray fluorescence and diffraction imaging techniques at NSLS and x-ray absorption near-edge structure analysis at Argonne National Laboratory's Advanced Photon Source (APS). Because the arsenic at mining sites may be in a pure form or part of a compound, and each soil sample might contain a mix of different forms of arsenic, each sample's detailed mineralogy varies, affecting its bioaccessibility.
"It's the work with the synchrotron that made this kind of experiment a success," said Reimer. "We couldn't have done it without the two synchrotrons."
In x-ray fluorescence imaging, researchers shoot x-rays at a sample, which absorbs the light and then emits another ray of light with a shifted wavelength. The wavelength depends on the energy levels of the sample, which vary from element to element and thus act as a unique fingerprint for a material. Measuring this wavelength gives the sample's energy levels, and thus its elemental makeup.
X-ray diffraction imaging, on the other hand, relies on x-rays that the sample does not absorb, but rather reflects. Detectors measure the scattered x-rays and use the pattern they form to recreate the structure of the sample.
X-ray absorption near-edge structure analysis at the APS allowed the researchers to determine the arsenic's oxidation state.
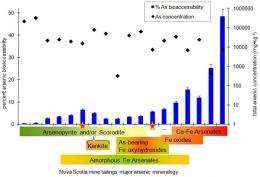
Next, researchers compared the mineralogical results from NSLS with the samples' bioaccessibility, and found some interesting connections.
Single arsenic minerals, such as arsenopyrite, had lower bioaccessibility — over 90 percent of the arsenic in the sample would pass harmlessly through the digestive system without being absorbed by the body. This is due to arsenopyrite's low solubility, or ability to dissolve in fluid. A more soluble compound, arsenic bearing iron(oxy)hydroxides, had 10 times the bioaccessibility of arsenopyrite.
Although these findings provide a new way to estimate bioaccessibility without running tests, this doesn't mean that those tests should be eliminated. For example, the compound yukonite (a calcium iron arsenate), which had the highest bioaccessibility of the tested minerals, could increase the bioaccessibility of the entire soil sample containing it. Despite knowing the bioaccessibility of the minerals in the sample, researchers still needed a bioaccessibility test of the entire sample to reveal its higher-than-expected potential toxicity.
"This has been quite an insight for us," Reimer said. "At the end of the day, you have to run the bioaccessibility test."
Nonetheless, mineralogical analysis has its own uses.
"Mineralogy is helpful in the case of unmixed minerals' bioaccessibility," Reimer said. In addition, the processes developed for this study can be applied to test the bioaccessibility of samples with a lower concentration of arsenic. Risk assessments can then include these results.
More information: L. Meunier, S.R. Walker, J. Wragg, M.B. Parsons, I. Koch, H.E. Jamieson, K.J. Reimer, "Effects of Soil Composition and Mineralogy on the Bioaccessibility of Arsenic from Tailings and Soil in Gold Mine Districts of Nova Scotia," Environ. Sci Technol., 44, 2667(2010).
Provided by Brookhaven National Laboratory