Graphene Outperforms Carbon Nanotubes for Creating Stronger, More Crack-Resistant Materials

(PhysOrg.com) -- Three new studies from researchers at Rensselaer Polytechnic Institute illustrate why graphene should be the nanomaterial of choice to strengthen composite materials used in everything from wind turbines to aircraft wings.
Composites infused with graphene are stronger, stiffer, and less prone to failure than composites infused with carbon nanotubes or other nanoparticles, according to the studies. This means graphene, an atom-thick sheet of carbon atoms arranged like a nanoscale chain-link fence, could be a key enabler in the development of next-generation nanocomposite materials.
“I’ve been working in nanocomposites for 10 years, and graphene is the best one I’ve ever seen in terms of mechanical properties,” said Nikhil Koratkar, professor in the Department of Mechanical, Aerospace, and Nuclear Engineering at Rensselaer, who led the studies. “Graphene is far superior to carbon nanotubes or any other known nanofiller in transferring its exceptional strength and mechanical properties to a host material.”
Results of Koratkar’s studies are detailed in three recently published papers: “Fracture and Fatigue in Graphene Nanocomposites,” published in Small; “Enhanced Mechanical Properties of Nanocomposites at Low Graphene Content,” published in ACS Nano; and “Buckling Resistant Graphene Nanocomposites,” published in the journal Applied Physics Letters.
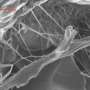
Advanced composites are increasingly a key component in the design of new windmill blades, aircraft, and other applications requiring ultra-light, high-strength materials. Epoxy composite materials are extremely lightweight, but can be brittle and prone to fracture. Koratkar’s team has infused the advanced composites with stacks, or platelets, of graphene. Each stack is only a few nanometers thick. The research team also infused epoxy composites with carbon nanotubes.
Epoxy materials infused with graphene exhibited far superior performance. In fact, adding graphene equal to 0.1 percent of the weight of the composite boosted the strength and the stiffness of the material to the same degree as adding carbon nanotubes equal to 1 percent of the weight of the composite. This gain, on the measure of one order of magnitude, highlights the promise of graphene, Koratkar said. The graphene fillers also boosted the composite’s resistance to fatigue crack propagation by nearly two orders of magnitude, compared to the baseline epoxy material.
Though graphene and carbon nanotubes are nearly identical in their chemical makeup and mechanical properties, graphene is far better than carbon nanotubes at lending its attributes to a material with which it’s mixed.
“Nanotubes are incredibly strong, but they’re of little use mechanically if they don’t transfer their properties to the composite,” Koratkar said. “A chain is only as strong as its weakest link, and if that link is between the nanotube and the polymer, then that is what determines the overall mechanical properties. It doesn’t matter if the nanotubes are super strong or super stiff, if the interface with the polymer is weak, that interface is going to fail.”
Koratkar said graphene has three distinct advantages over carbon nanotubes. The first advantage is the rough and wrinkled surface texture of graphene, caused by a very high density of surface defects. These defects are a result of the thermal exfoliation process that the Rensselaer research team used to manufacture bulk quantities of graphene from graphite. These “wrinkly” surfaces interlock extremely well with the surrounding polymer material, helping to boost the interfacial load transfer between graphene and the host material.
The second advantage is surface area. As a planer sheet, graphene benefits from considerably more contact with the polymer material than the tube-shaped carbon nanotubes. This is because the polymer chains are unable to enter the interior of the nanotubes, but both the top and bottom surfaces of the graphene sheet can be in close contact with the polymer matrix.
The third benefit is geometry. When microcracks in the composite structure encounter a two-dimensional graphene sheet, they are deflected, or forced to tilt and twist around the sheet. This process helps to absorb the energy that is responsible for propagating the crack. Crack deflection processes are far more effective for two-dimensional sheets with a high aspect ratio such as graphene, as compared to one-dimensional nanotubes.
Koratkar said the aerospace and wind power industries are seeking new materials with which to design stronger, longer-lived rotor and wind turbine blades. His research group plans to further investigate how graphene can benefit this goal. Graphene shows great promise for this because it can be produced from graphite, which is available in bulk quantities and at relatively low cost, he said, which means mass production of graphene is likely to be far more cost effective than nanotubes.
Co-authors on the three papers include Rensselaer mechanical engineering graduate students Mohammed A. Rafiee, Javad Rafiee, and Iti Srivastava; as along with Professor Zhong-Zhen Yu’s group at the Beijing University of Chemical Technology.
More information: For more information on Koratkar’s research, visit: www.rpi.edu/~koratn .
Provided by Rensselaer Polytechnic Institute