Cow Brain Protein May Hold Alternative Energy Promise

(PhysOrg.com) -- Of all the ideas that hold promise in alternative energy, cow brains are an odd candidate. They do not fit into the list of usual plant-based subjects, such as corn or switch grass. But cow brains contain an abundance of clathrin, an important protein in cellular biology, regenerative medicine and perhaps even alternative energy.
Dr. Sarah Heilshorn of the Stanford Institute of Materials and Energy Science, a joint SLAC and Stanford venture, is interested in investigating the capabilities of this nifty protein in collaboration with SIMES colleagues Nick Melosh, Andy Spakowitz and Seb Doniach. The scientists are curious how clathrin might be used to form nanostructured inorganic materials for applications in batteries and solar cells. This protein can essentially be used as a skeletal template for the growth of inorganic materials into precise structures. Once scientists assemble the clathrin protein into the proper structures, they can attach other atoms and molecules, much like adding windows and walls to the frame of a house.
Heilshorn and her team are interested in the fundamental questions of how these proteins assemble, with the goal of beginning to tap into biology's secrets for creating complex molecules. Cells have a much better knack for building tiny structures than humans do. For example, viruses have been used quite a bit as a template for building inorganic materials such as electrodes for batteries. Heilshorn and her colleagues were interested in finding more biological templates, for a wider range of uses.
Despite its important role in physiology, clathrin's potential as a template only emerged very recently. Scientists noted that clathrin could form structures outside its natural environment back in the 1980s, though it was merely thought of as curious but not particularly useful.
"When I saw the beautiful structures that clathrin could form, I started to think maybe it could be a really nice template," Heilshorn said.
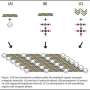
Clathrin exists in every cell in the human body, as an important gate-keeper. The protein is shaped like a tripod: three spindly legs joined together at one hub. Clathrin molecules easily combine with each other, forming a honeycomb-like lattice on the inner surface of cells. When the right molecules attach themselves to clathrin, the lattice structure buckles into the cell, forming a pouch. This pouch pinches off the cell membrane, delivering its molecular cargo to the cell's interior.
Clathrin's role in cell transport is a complicated biochemical process, relying on just the right triggers. But surprisingly, it is not that difficult to get clathrin to form structures outside of the cell. First, scientists extract clathrin from tissue, such as cow brain. With adjustments to the pH, clathrin concentration and salt concentration, clathrin will assemble itself into a useful skeletal structure. In a test tube, it can form cubes, spheres, barrels and tetrahedra, among other shapes.
"Nature has evolved all these great ways to build inorganic materials with amazing control over shape and structure," Heilshorn said. Even more, these biological structures are created at room temperature and pressure. Production of synthetic materials typically relies on harsh chemicals, very high temperatures and pressures.
These clathrin structures provide a skeleton onto which scientists can add inorganic atoms and molecules, creating materials such as catalysts and electrodes. Since the clathrin protein has a very precise pattern of positive and negative charges decorating its surface, the Heilshorn group can engineer strategies to bond various inorganic molecules at specific locations on the template surface. So far, the group has successfully incorporated gold and titanium dioxide, also known as "titania." In the future, they will also look to use platinum and cobalt oxide.
Such materials, assembled on a nanoscopic scale, could make excellent batteries and solar cells. Titania, for example, has photocatalytic properties and can absorb sunlight to split water into oxygen and hydrogen. By creating these inorganic materials using clathrin, scientists can tailor their structure toward converting chemicals and sunlight into energy.
"This is something I wouldn't have started if I wasn't part of SIMES," Heilshorn said. Her research group had been interested in cellular and medical applications of protein structures, but because of SIMES' focus on energy, they saw a multidisciplinary opportunity. Now SIMES adds yet another dimension, albeit an unusual one, to the search for cleaner energy.
Provided by SLAC National Accelerator Laboratory