Argonne scientists seek natural remediation for uranium-rich sites

While most of us are focused on life above ground, scientists at the U.S. Department of Energy’s (DOE) Argonne National Laboratory are trying to understand the drama unfolding beneath our feet.
Their work centers on the more than billions of tons of bacteria living within the Earth’s subsurface, below the root zone, and how they change the chemical composition of the rocks and minerals they touch, including uranium. The result could prove useful in a surprising way.
Scientists know some bacteria can change Uranium (VI)—which is radioactive, toxic and water-soluble—into the more insoluble, stationary and thus less harmful Uranium (IV).
What they don't know is how to harness this chemical transformation so it could help reduce the danger posed by the more than 1,000 uranium-contaminated sites scattered across the nation.
Some of these sites—relics of the Cold War era—are near major waterways, including the Colorado, Tennessee and Columbia Rivers, making their remediation a high priority for the DOE.
Uranium (VI) can be transported by groundwater, so wherever water goes, the uranium goes with it. The key is to make it immobile, to stop it before it's introduced into the food chain and ultimately to humans.
Certain species of bacteria can do just that.
"We know that some bacteria turn uranium into rock form so that it can't move," said Ken Kemner, an Argonne physicist. "That could be critical for some of these sites.
But the process isn't without a few kinks. That's because uranium isn't the only element that bacteria can change—and this is where a seemingly simple process grows complicated.
"Bacteria cause chemical changes in several elements, including iron and sulfur," Kemner said. "And those changes in the iron and/or sulfur geochemistry could cause the uranium to change between soluble and insoluble forms."
It's these problems that Kemner and a team of Argonne microbiologists, chemists, physicists and geochemists are trying to figure out. They're working to find a way to enhance the stability of the immobile form of uranium while many other processes are occurring.
"We want to know why and how this happens, so that we might one day use this process to our advantage in terms of cleaning up sites contaminated with heavy metals and radioactive elements," Kemner said.
The Argonne team is part of the DOE’s efforts in Subsurface Biogeochemical Research, which performs some of the basic research needed to tackle some of the country's most challenging environmental cleanup problems.
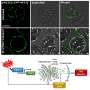
Argonne scientists are uniquely positioned because they have access to the laboratory's Advanced Photon Source (APS). The APS provides the brightest x-ray beams in the Western Hemisphere, allowing scientists to understand uranium's transformation at the atomic level.
Argonne researchers recently published two papers on the topic. Both appeared in the journal Environmental Science and Technology, and both focused on uranium immobilization. The first was published in January 2010 and the second a month later.
In the first study, Argonne's team used the APS to better understand the chemical transformations of uranium and iron. In the second study, scientists used the APS to investigate how iron helps to chemically transform and immobilize uranium underground.
While Kemner and his associates conduct much of their research in the laboratory, they are also part of research teams that work out in the field. The teams study bacteria at three uranium-contaminated research sites across the country: one in Colorado and two others at Pacific Northwest and Oak Ridge National Laboratories.
Their findings are promising.
"The Department has successfully demonstrated in some pilot studies that this really works," Kemner said. "But we don't understand it enough that we can go off into the environment and routinely employ the bacteria on a larger scale."
Kemner emphasizes that the research is not focused on creating new bacteria and releasing them into the environment, as some critics have feared.
"We are working only with the indigenous population," he said. "We have all the bacteria we need underground to do the job."
Argonne's most recent papers on this topic are available online from the journal Environmental Science and Technology, here and here.
Provided by Argonne National Laboratory