Measuring the Speed of Noble 'Bubbles'
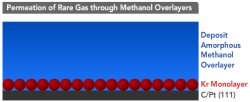
(PhysOrg.com) -- Using a layer of noble gas "bubbles," scientists at Pacific Northwest National Laboratory devised a straightforward way to measure how fast molecules diffuse in supercooled liquids. Working at temperatures far below room temperature, the scientists placed a layer of krypton or argon atoms under the liquid, which was chilled to within a few degrees of freezing solid. Then, they measured how fast the atoms bubbled up to surface. Using this data, they calculated the diffusion through larger or macroscopic samples. To be able to do this, they used liquid films that were only 10-100 nanometers thick.
"Diffusion is trillions of times slower at these temperatures than at room temperature," said Dr. Scott Smith, an experimental chemist on the project. "Diffusion studies at this low temperature couldn't be done on the macroscopic scale. There simply weren't the eons available."
From energy usage and storage in batteries to pharmaceutical shelf life, understanding the structure and stability of supercooled liquids and the amorphous solids they freeze into could address many open questions. This new technique provides answers at temperatures around 100 to 115K. Previously, the lowest temperature for which diffusion data could be obtained for methanol which was studied here was 252K.
"The method dramatically increases both the temperature and dynamic range of the diffusion measurements," said Dr. Bruce Kay, a leading experimental chemical physicist at PNNL.
Imagine you've just entered a very crowded party. You want to get to the door on the other side of the room. Your movement through the crowd is somewhat related to how fast the other people in the room are moving. If they are packed close together and not moving, it will take longer to get to your destination. If the dense crowd is moving, you'll reach your destination faster. In a similar way, the scientists showed that the movement of atoms of inert or noble gas through a supercooled liquid is related to the movement of the liquid molecules.
Their new straightforward approach began with the scientists putting a layer of krypton atoms under the liquid to be tested. They then covered this layer of atoms with the molecules they wanted to study. All this was done at low temperatures using molecular beam techniques. Then, they applied heat. This caused the molecules on top to start moving so the krypton atoms could bubble through the material. The researchers could not track the progress of the krypton moving around inside the material. However, using a mass spectrometer, they measured when the atoms reached the material's surface. This data was used to calculate the diffusion rate.
The scientists are applying this new technique to other glassy materials and other ‘bubble' atoms to learn more about materials of interest to the Department of Energy and other clients.
"This is an easy way to get some understanding of molecules," said Dr. Jesper Matthiesen, an experimental chemical physicist and postdoctoral fellow at PNNL. "This method is fast, easy, and versatile."
More information: Matthiesen J, RS Smith, and BD Kay. 2009. "Using Rare Gas Permeation to Probe Methanol Diffusion near the Glass Transition Temperature." Physical Review Letters 103(24): 245902-1 - 245902-4. DOI:10.1103/PhysRevLett.103.245902
Provided by Pacific Northwest National Laboratory