Researchers take early step towards devising new antibiotics
(PhysOrg.com) -- Researchers have made the first step towards creating a new class of antibiotics in a study published last night in the journal Proceedings of the National Academy of Sciences.
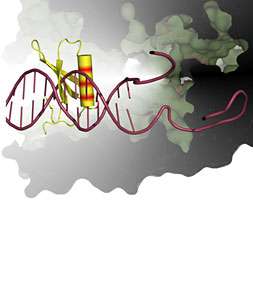
The scientists, from Imperial College London, have revealed the structure of a protein called Gp2. This protein is produced by the T7 virus, which only targets bacteria and which infects and kills them by interfering with the way in which they express genes.
Gp2 works by interfering with an essential enzyme in bacteria called the RNA Polymerase, which represents the central machinery that enables information contained in their genes to be “read”. If the RNA Polymerase is unable to function, the bacteria cannot survive. Today’s is the first study to show the shape of a protein that is able to strongly inhibit RNA Polymerase and is derived from a virus.
The researchers behind today’s study hope that ultimately it will be possible to develop new drugs that mimic the structure and mode of action of Gp2 in order to combat bacterial infections.
There is already a group of clinically used antibiotics, known as rifamycin, which interfere with the RNA polymerase from bacteria, but against which many bacteria have developed resistance. Accordingly, the researchers hope that if new drugs can be devised that mimic Gp2, these will be less susceptible than existing antibiotics to antibiotic resistance, because they will work in a different way to the antibiotics that are currently available.
The next step for this research will be for scientists to identify small molecule mimics of Gp2 that can be used to create these drugs.
Today’s findings were the result of an interdisciplinary collaboration between researchers from two cross-faculty groups at Imperial - the Centre for Molecular Microbiology and Infection (CMMI) and the Centre for Structural Biology (CSB). The corresponding authors of the study, Dr Sivaramesh Wigneshweraraj from the Department of Microbiology in Medicine, together with Professor Stephen Matthews and Dr Ernesto Cota from the Department of Life Sciences in Natural Sciences, also worked closely with colleagues at Rutgers University in New Jersey, USA, and the Russian Academy of Science.
Dr Sivaramesh Wigneshweraraj, who conducted the analyses of how Gp2 interferes with the intricate functionalities of the bacterial RNA polymerase, said: “This work is at a very early stage but knowing the shape of viral proteins like Gp2 and how they operate represents an important first step towards creating new drugs against bacteria from viruses that infect and kill bacteria”.
Professor Steve Matthews, from the Centre for Structural Biology, whose group calculated the structure of the protein, said: ”Our collaboration not only provides the first atomic resolution insights into this potent inhibitor, but also provide a useful blueprint for future collaborations between the CSB and CMMI aimed at combating bacterial infections”
Provided by Imperial College London
















