Imaging a catalyst one atom at a time

(PhysOrg.com) -- The catalytic processes that facilitate the production of many chemicals and fuels could become much more environmentally friendly thanks to a breakthrough achieved by researchers from Lehigh and Rice Universities.
In an article published Nov. 8 by the journal Nature Chemistry, the researchers report a novel electron microscopy imaging study of a tungstated zirconia solid acid catalyst. Based on new information obtained from these images, the researchers were able to design a preparation procedure that increased the activity of the catalyst by more than 100 times.
A catalyst is a substance that accelerates the rate of a chemical reaction without itself being consumed by that reaction. Liquid acid catalysts are widely used in the production of many chemicals but pose environmental concerns due to evaporation, spilling and corrosion. Chemical companies are seeking to replace liquid catalysts with solid acid catalysts, which can be more cleanly used and disposed of because they do not evaporate, spill or lead to corrosion.
The Lehigh-Rice team used aberration-corrected scanning transmission electron microscopy (STEM) and advanced optical microscopy and spectroscopy techniques, including Raman, infrared and ultraviolet-violet visible spectroscopies, to shed light on the nanostructure and nanoscale behavior of a tungstated zirconia solid acid catalyst. Among other applications, tungstated zirconia is used to improve the octane content of gasoline through a process called isomerization, in which a straight chain alkane molecule is converted into a branched chain molecule.
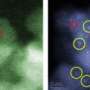
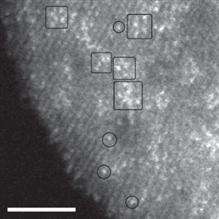
The team was able to directly image a variety of tungsten-oxide species, including monomers, polymer-like chains and sub-nanometer clusters, which were supported on a nanocrystalline zirconia substrate. Catalytic performance studies revealed that the most active catalytic species are tungsten oxide clusters that measure only 0.8 to 1 nm in diameter and are mixed with a few zirconium atoms emanating from the support. A nanometer is one-billionth of a meter, or about the diameter of 10 hydrogen atoms.
The team then deliberately deposited the catalytically active sub-nanometer mixed tungsten-zirconium oxide clusters onto a tungstated zirconia catalyst that previously had possessed low catalytic activity. When the catalytic activity of the poor catalyst was found to have improved by two orders of magnitude, the team's hypothesis about the identity and structure of the active species within the tungstated zirconia material was confirmed. The researchers have filed a patent application for their new catalyst preparation method.
The Nature Chemistry article, titled "Identification of active Zr-WOx clusters on a ZrO2 support for solid acid catalysts," has six authors. Wu Zhou, the lead author, is a Ph.D. candidate in materials science and engineering at Lehigh. The other authors are Elizabeth Ross-Medgaarden, who earned a Ph.D. in chemical engineering from Lehigh in 2007; William V. Knowles, who earned a Ph.D. in chemical and biomolecular engineering from Rice in 2006; Michael S. Wong, associate professor of chemical and biomolecular engineering and of chemistry at Rice; Israel E. Wachs, professor of chemical engineering at Lehigh; and Christopher J. Kiely, professor of materials science and engineering at Lehigh, who is also the corresponding author of the paper.
The research was funded by the National Science Foundation through its Nanoscale Interdisciplinary Research Team (NSF-NIRT) program. Wachs, who directs Lehigh's Operando Molecular Spectroscopy and Catalysis Laboratory, is principal investigator on the grant. Kiely, director of Lehigh's Nanocharacterization Laboratory, is co-principal investigator, as is Wong, who directs the Catalysis and Nanomaterials Laboratory at Rice.
The Lehigh-Rice team also collaborates closely with Matthew Neurock, a professor of chemical engineering and an expert in computational and theoretical catalysis at the University of Virginia. Neurock is a co-PI on the NSF-NIRT project.
A new view of an old problem
The Lehigh-Rice team attributes much of its success to the use, for the first time on tungstated zirconia catalysts, of aberration-corrected scanning transmission electron microscopy (STEM) and its integration with three optical spectroscopic techniques—Raman, infrared and ultraviolet-visible. Only by combining the microscopy and spectroscopy studies, says Wachs, was it possible to obtain the insights at the molecular level that are required to pinpoint the origin of the acidity of the tungstated zirconia.
Lehigh four years ago became the first university in the world to acquire two aberration-corrected STEM instruments. The VG HB 603 STEM can map the chemical composition of nanoparticles, while the JEOL 2200 FS STEM has unparalleled imaging capabilities. The researchers employed a microscopy technique called high-angle annular dark-field imaging (HAADF), which uses a focused beam of electrons only 1 angstrom (0.1 nm) wide, to obtain clear images of the supported tungsten oxide species.
"HAADF imaging of tungstated zirconia catalysts in an aberration-corrected STEM allows, for the first time, direct imaging of the various [catalytic] species present," the researchers wrote in Nature Chemistry.
Wachs, who is internationally renowned for his work in catalysis and his expertise in Raman and other spectroscopy techniques, especially under reaction conditions, said the aberration-corrected STEMs has opened an unprecedented window on the structure and size of the catalytic species.
"This new generation of aberration-corrected STEMs enables us finally to see the dimensions of the species we're studying," says Wachs. "We can see monomers, dimers and trimers, as well as larger clusters of tungsten oxide."
The researchers used information gleaned from the aberration-corrected STEM studies in concert with data obtained from Raman, IR and UV-visible spectroscopy, and from controlled catalyst testing studies, to molecularly engineer a better catalyst, says Wachs. The spectroscopy experiments were performed in situ as catalysis was occurring over the tungstated zirconia.
Wachs' Operando Molecular Spectroscopy and Catalysis Laboratory is home to the most advanced optical spectroscopic instrumentation in catalysis in America. The high resolution-Raman spectrometer (Horiba Scientific LabRaman-HR) is integrated with IR and UV-visible spectroscopy in one system to enable the contemporaneous collection of multiple spectroscopic information from the same catalyst spot. The optical techniques also function under reaction conditions (gas-solid and aqueous-solid) and the effluent reaction products from the catalytic reactor cell are simultaneously monitored with mass spectrometry. All information is collected in real time (nanosecond to second range).
"The combination of these imaging and spectroscopy techniques enabled us to make a an active catalytic site, deposit it on a catalyst with low activity, and show a 100-fold improvement in catalytic activity," says Wachs. "In short, we've been able to design, on demand, the active catalytic sites by molecularly engineering the catalyst.
"These imaging and specstroscopy techniques are very complementary. They are like multiple pairs of eyes that help us see what is going on at the atomic and molecular scale during the catalytic reaction."
Clearing up a mystery
Lehigh's STEM instruments are equipped with spherical aberration correctors that improve imaging and chemical mapping resolution by overcoming distortions in the lenses that focus the electron beams onto the specimen. This improved resolution enables researchers to view individual atoms, especially of heavy elements like tungsten.
"Using conventional high resolution electron microscopy, it is next to impossible to see individual tungsten atoms on the zirconia substrate," says Zhou. "If you can correct the spherical aberration in the microscopy, this allows you in HAADF mode to pick up the heavy tungsten atoms, which show up as bright specks against the lighter zirconia substrate."
The ability to visualize individual supported atoms, Zhou adds, has helped solve a mystery that has puzzled scientists since the tungstated zirconia catalyst was developed two decades ago by researchers in Japan, namely, which specific structural feature of the catalyst is responsible for its catalytic activity?
Being able to see individual tungsten atoms allowed the researchers to identify the active catalytic sites in these solid acid catalysts. The team took a set of samples, some with low catalytic activity, some with high activity, and compared their nanostructures. In both sets of samples, they found isolated monomers and linked chains of polymeric tungstate species, which turn out to have little catalytic activity.
"Only in the samples with high catalytic activity," says Zhou, "did we find 3-D mixed zirconium-tungsten oxide clusters, less than 1 nm in size, which are the active catalytic sites in these solid acid catalysts."
"By identifying the nano feature that is responsible for the desired catalytic performance," says Wong, who is an expert in catalyst synthesis and materials chemistry, "we can then focus research efforts on rationally designing new ways to prepare catalytic materials with only that particular feature."
Kiely published a related catalysis paper in Science last fall in which he reported that individual atoms of gold on a metal oxide were imaged using the aberration-corrected HAADF method.
"It has already been established that the dispersion of a metal on a metal-oxide support could be seen with aberration-corrected electron microscopy," he says. "This new Nature Chemistry study is the first report of the use of aberration-corrected microscopy to image the atomic structure of a metal oxide overlayer deposited onto a metal-oxide support."
The Lehigh-Rice NIRT team published an article earlier this year in the Journal of the American Chemical Society that describes how the integration of microscopy and spectroscopy allowed for the tuning of the electronic and molecular structures of nanostructured catalytic active sites for acid and redox reactions. This integration is now being applied to the molecular engineering of catalysts used in the production of clean liquid fuels and in the manufacture of agents that control power plant emissions responsible for acid rain.
Source: Lehigh University (news : web)