Scientists harness nanoparticles to track cancer-cell changes
The more dots there are, the more accurate a picture you get when you connect them. A new imaging technology could give scientists the ability to simultaneously measure as many as 100 or more distinct features in or on a single cell. In a disease such as cancer, that capability would provide a much better picture of what's going on in individual tumor cells.
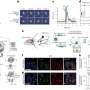
A Stanford University School of Medicine team led by Cathy Shachaf, PhD, an instructor in microbiology and immunology, has for the first time used specially designed dye-containing nanoparticles to simultaneously image two features within single cells. Although current single-cell flow cytometry technologies can do up to 17 simultaneous visualizations, this new method has the potential to do far more. The new technology works by enhancing the detection of ultra-specific but very weak patterns, known as Raman signals, that molecules emit in response to light.
In a study to be published April 15 in the online journal PLoS-ONE, the Stanford team was able to simultaneously monitor changes in two intracellular proteins that play crucial roles in the development of cancer. Successful development of the new technique may improve scientists' ability not only to diagnose cancers — for example, by determining how aggressive tumors' constituent cells are — but to eventually separate living, biopsied cancer cells from one another based on characteristics indicating their stage of progression or their degree of resistance to chemotherapeutic drugs. That would expedite the testing of treatments targeting a tumor's most recalcitrant cells, said Shachaf, a cancer researcher who works in a laboratory run by the study's senior author, Garry Nolan, PhD, associate professor of microbiology and immunology and a member of Stanford's Cancer Center.
Cancer starts out in a single cell, and its development is often heralded by changes in the activation levels of certain proteins. In the world of cell biology, one common way for proteins to get activated is through a process called phosphorylation that slightly changes a protein's shape, in effect turning it on.
Two intracellular proteins, Stat1 and Stat6, play crucial roles in the development of cancer. The Stanford team was able to simultaneously monitor changes in phosphorylation levels of both proteins in lab-cultured myeloid leukemia cells. The changes in Stat1 and Stat6 closely tracked those demonstrated with existing visualization methods, establishing proof of principle for the new approach.
While the new technology so far has been used only to view cells on slides, it could eventually be used in a manner similar to flow cytometry, the current state-of-the-art technology, which lets scientists visualize single cells in motion. In flow cytometry, cells are bombarded with laser light as they pass through a scanning chamber. The cells can then be analyzed and, based on their characteristics, sorted and routed to different destinations within the cytometer.
Still, flow cytometry has its limits. It involves tethering fluorescent dye molecules to antibodies, with different colors tied to antibodies that target different molecules. The dye molecules respond to laser light by fluorescing — echoing light at exactly the same wavelength, or color, with which they were stimulated. The fluorescence's strength indicates the abundance of the cell-surface features to which those dyes are now attached. But at some point, the light signals given off by multiple dyes begin to interfere with one another. It is unlikely that the number of distinct features flow cytometry can measure simultaneously will exceed 20 or so.
The new high-tech dye-containing particles used by the Stanford team go a step further. They give off not just single-wavelength fluorescent echoes but also more-complex fingerprints comprising wavelengths slightly different from the single-color beams that lasers emit. These patterns, or Raman signals, occur when energy levels of electrons are just barely modified by weak interactions among the constituent atoms in the molecule being inspected.
Raman signals are emitted all the time by various molecules, but they're ordinarily too weak to detect. To beef up their strength, the Stanford team employed specialized nanoparticles produced by Intel Corp., each with its own distinctive signature. Intel has designed more than 100 different so-called COINs, or composite organic-inorganic nanoparticles: These are essentially sandwiches of dye molecules and atoms of metals such as silver, gold or copper whose reflective properties amplify a dye molecule's Raman signals while filtering out its inherent fluorescent response. The signals are collected and quantified by a customized, automated microscope.
Shachaf anticipates being able to demonstrate simultaneous visualization of nine or 10 COIN-tagged cellular features in the near future and hopes to bring that number to 20 or 30, a new high, before long. "The technology's capacity may ultimately far exceed that number," she added. Some day it could be used for more than 100 features. Meanwhile, another group outside Stanford, now collaborating with the Nolan group, has developed a prototype device that can detect Raman signals in a continuous flow of single cells, analogous to flow cytometry but with higher resolving power, Shachaf said.
Source: Stanford University Medical Center (news : web)