November 7, 2008 feature
Scientists revisit 1833 hydrogen production experiment
(PhysOrg.com) -- In the early 1800s, during the peak of the Industrial Revolution, modern science revolved around steam engines and other coal-powered applications. So it may seem a bit out of place that, in 1833, an Italian physicist named G. D. Botto was performing experiments on a technique for generating hydrogen.
“Hydrogen is the most common element on Earth, the lightest and the simplest in its structure,” Roberto De Luca of the University of Salerno in Italy told PhysOrg.com. “Hydrogen gas was already known to [French chemist Antoine-Laurent] Lavoisier at the end of the eighteenth century. However, the production of hydrogen, at the time Botto performed his experiment, can only be viewed as a sub-product of electrolysis. Botto’s main objective was to show to the scientific community that electricity could be obtained by a source of heat through his ingenious device.”
De Luca is part of an Italian team of scientists who have revisited Botto’s experiments to investigate whether the technique could have applications for today’s energy problems. The Italian group was inspired by the convenience of Botto’s device, which can be easily fabricated and is composed of widely available materials. The researchers calculated that, although a modified version of Botto’s device has a very low power conversion efficiency, it can still produce enough electromotive force to generate hydrogen.
“We learned about Botto’s idea through the invaluable work of Professor [Salvatore] Ganci [also a co-author of this study], who is an expert in the history of physics,” De Luca said. “He is now writing a book on scientific instruments constructed by physicists from the province of Genoa in the early 19th century. Among his studies, there is the device used by Botto, by which it was possible to prove that electricity could be generated by heating an array of thermocouples. Without the invaluable contribution of Professor Ganci, this work would have not been possible.”
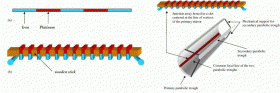
Botto’s original 19th-century device is quite simple. It consists of a chain of iron and platinum wires alternately connected as thermocouples, which are used to convert a temperature difference into an electric voltage. Botto then wrapped the chain around a wooden stick so that the iron-platinum junctions were evenly positioned on opposite sides of the stick. By heating the contraption with a flame of burning alcohol, Botto could generate an electromotive force. Further, by passing the generated electric current through water, he demonstrated how the method could be used to produce hydrogen through electrolysis.
The Italian team studied Botto’s device, but made some significant adjustments. They first considered substituting copper for platinum in the thermocouples. They also proposed completely replacing Botto’s thermocouples with thermoelectric semiconductors for greater efficiency.
Also, rather than use a flame of burning alcohol as the heat source, the Italian team considered using solar power to heat the thermocouples/semiconductors and create a temperature difference. They suggested that two parabolic mirrors could be arranged to concentrate solar radiation on the thermocouples and heat one side. To cool the other side (in order to create a temperature difference), the wooden stick might be replaced with a hollow electrically insulating material through which water could run to cool the desired junctions.
The researchers then estimated the temperature difference, which could be used to estimate the voltage difference, which was only about 1 mV. They also estimated a small power output of about 20 mW. Despite the low power conversion efficiency, the Italians’ proposed solar-powered device could generate enough current to produce hydrogen gas through electrolysis.
“We think that this idea can be used in the production of hydrogen gas directly from solar energy, through electrolysis,” De Luca said. “However, nowadays, one would not use thermocouples, as in Botto’s experiment, but could, more efficiently, use thermoelectric semiconductors to obtain a much higher power output. The important point in this work is also that, while there has always been solid scientific ground for these ideas, there has also been a lot of resilience in their applications.”
As for why the researchers suggested using solar radiation to heat the device rather than a flame, De Luca emphasized the economic benefits. He and others have investigated unique, simple methods for capturing sunlight in other studies, and explained the advantages of solar energy.
“It makes a great difference, in hydrogen production, to use free energy from a nuclear power plant which starts working early in the morning and stops early at night,” he said, referring to our Sun. “This energy will be free for the next five billion years, approximately. Hydrogen can thus be freely produced (unless we consider the cost of water) as long as this wonderful, perfectly clean power plant will be shining upon us. Hydrogen can then be used to run our cars and to make our houses bright at night in the future, despite the fact that Earth might sooner or later run out of oil.”
More information: De Luca, R.; Ganci, S.; and Zozzaro, P. “Revisiting an idea of G D Botto: a solar thermoelectric generator.” Eur. J. Phys. 29 (2008) 1295-1300.
Copyright 2008 PhysOrg.com.
All rights reserved. This material may not be published, broadcast, rewritten or redistributed in whole or part without the express written permission of PhysOrg.com.