March 7, 2008 feature
Quantum Dots May Lead to Rainbow Solar Cell
For the first time, researchers have created solar cells made of different-sized quantum dots, each tuned to a specific wavelength of light. By arranging these quantum dots in an ordered pattern, the scientists hope that they can one day fabricate “rainbow” solar cells, which can efficiently harvest a large part of the useful spectrum of sunlight.
The group of researchers from the University of Notre Dame, Anusorn Kongkanand, Kevin Tvrdy, Kensuke Takechi, Masaru Kuno, and Prashant Kamat, have published their study in a recent issue of the Journal of the American Chemical Society. Their research was funded by the Office of Basic Energy Sciences of the Department of Energy.
Using quantum dots to absorb light has a unique advantage over other light-absorbing materials: the “size quantization effect.” By varying the size of the tiny semiconductor quantum dots, the researchers can tune the solar cells to absorb light of certain wavelengths. Smaller quantum dots absorb shorter wavelengths of light, while larger quantum dots absorb longer wavelengths.
By combining different-sized quantum dots on one solar cell, the researchers can create solar cells that absorb more light and thereby deliver power at greater efficiencies compared with solar cells made of bulk semiconductors.
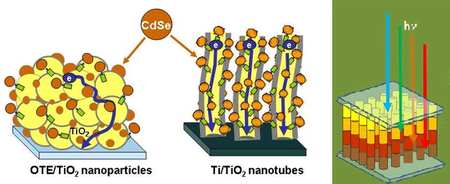
In the Notre Dame study, the scientists assembled cadmium selenide (CdSe) quantum dots in a single layer on the surface of nano films and tubes made of titanium dioxide (TiO2). After absorbing light, the quantum dots inject electrons into the TiO2 structures, which are then collected at a conducting electrode that generates photocurrent.
“Anchoring CdSe quantum dots on TiO2 nanotubes allowed us to create an ordered assembly of nanostructures,” Kamat told PhysOrg.com. “This architecture facilitated efficient transport of electrons to the collecting electrode surface and allowed us to achieve efficiency improvement.”
The researchers used four different sizes of quantum dots (between 2.3 and 3.7 nm in diameter) which exhibited absorbent peaks at different wavelengths (between 505 and 580 nm). The group observed a trade-off in performance corresponding with quantum dot size: smaller quantum dots could convert photons to electrons at a faster rate than larger quantum dots, but larger quantum dots absorbed a greater percentage of incoming photons than smaller dots. The 3-nm quantum dots offered the best compromise, but the researchers plan to improve both the conversion and absorption performances in future prototypes.
Besides investigating the quantum dots’ size quantization effect, the researchers also experimented with two different nano architectures – particle films and nanotubes – that act as scaffolds for transporting electrons from the quantum dots to the electrodes. The group found that the hollow 8000-nm-long nanotubes, where both the inner and outer surfaces were accessible to quantum dots, could transport electrons more efficiently than films.
With the development of the first solar cell with multi-sized quantum dots, the researchers plan to take the next steps and design rainbow solar cells, which would contain different-sized quantum dots assembled in an orderly fashion. With small quantum dots on the outer edge of the cell absorbing blue light, the red light (with longer wavelengths) would pass through the outer layer but still be absorbed by larger quantum dots located in the inner layer. This “rainbow” gradient would combine the faster electron injection rate of small quantum dots and greater absorption range of larger quantum dots, and ultimately lead to a highly efficient solar cell.
“Usually, silicon-based photovoltaic panels operate with an efficiency of 15-20%,” Kamat said. “Silicon solar cells generate only one electron-hole pair per incident photons, irrespective of their energy. Thus, the higher energy of blue light is simply wasted in terms of heat. The obvious question is, can nanotechnology provide new ways to harvest these higher energy photons more efficiently?
“Semiconductor quantum dots seem to be the answer. They are capable of producing multiple charge carriers when excited with high energy light. If we succeed in capturing these charge carriers, we can expect significantly higher efficiencies. The target is to reach efficiency values greater than 30% using quantum dot rainbow solar cells.”
To achieve this efficiency, Kamat explained that there are two main challenges. The first is organizing the light harvesting nanostructures so that they efficiently absorb light in the visible and near infrared region, and transport electrons within the films. Secondly, the quantum dots should generate multiple charge carriers to be captured to generate photocurrent.
“New advances in nanotechnology are key to the success of developing highly efficient and cost-effective solar cells,” he said.
Home owners can attach solar cells to roofs, of course. But, as the Notre Dame researchers suggest, rainbow solar cells might also be used to develop colored windows, where the color can be tuned by changing the size of the quantum dots. Just as in rooftop cells, the quantum dots in the glass could absorb light that could then be converted into electricity.
More information: Kongkanand, Anusorn; Tvrdy, Kevin; Takechi, Kensuke; Kuno, Masaru; and Kamat, Prashant V. “Quantum Dot Solar Cells. Tuning Photoresponse through Size and Shape Control of CdSe-TiO2 Architecture.” J. Am. Chem. Soc. March 1, 2008. DOI: 10.1021/ja0782706.
Copyright 2008 PhysOrg.com.
All rights reserved. This material may not be published, broadcast, rewritten or redistributed in whole or part without the express written permission of PhysOrg.com.