Melting snow provides clues for acidification
In forests of the northeastern United States, sulfate and nitrate are the dominant dissolved forms of sulfur and nitrogen in precipitation. In winter, these acidic agents accumulate in the snowpack and are released to groundwater and streams over a short period of time during spring snowmelt. This pulsed release of sulfate and nitrate in snowmelt can cause episodic acidification in poorly buffered soils, ultimately threatening the health of acid-sensitive biota.
There have been recent studies showing that biological cycling of sulfur and nitrogen persists in cold weather, despite below freezing air temperatures. Much of this activity occurs in soils, where an insulating snow layer keeps soil temperatures warm enough for a range of biological processes. Despite the growing awareness of winter’s role in sulfur and nitrogen cycling, many questions remain unanswered. In particular, there is much uncertainty about how sulfate and nitrate are retained or transformed in forest soils during cold weather.
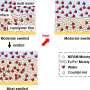
In the November-December 2007 issue of the Soil Science Society of America Journal (SSSAJ), scientists from the U.S. Forest Service, SUNY-ESF, University of Calgary, and Cary Institute of Ecosystem Studies tracked the movement of sulfate and nitrate deposited in snow. A solution containing isotopically enriched sulfate and nitrate was sprayed on the surface of the snowpack during mid winter. The isotopic values of the labeled sulfate and nitrate were well above background levels and served as a tracer to follow the movement and transformation of these compounds in the ecosystem.
The researchers found that almost all of the labeled sulfate and nitrate deposited on the surface of the snow was recovered in snowmelt water, indicating that there were no significant transformations of sulfate and nitrate in the snowpack.
In contrast, about half of the sulfate and nitrate was retained or transformed in the forest floor, suggesting that organic soils are a sink for these compounds during winter. For sulfate, the amount retained or transformed in the forest floor was nearly equal to the amount released, resulting in no significant net gains or losses. A significant amount of ammonium was produced in the forest floor indicating that N mineralization can be important, even when soil temperatures are near freezing. By contrast, net nitrification rates were very low during winter.
Tracer results indicated that microbes did not immobilize snowpack nitrate and that other processes such as plant uptake, denitrification, and abiotic nitrate retention were probably more important factors affecting nitrate during snowmelt. More information on controls on nitrogen and sulfur cycling during winter is critical to our understanding of long-term trends and will help us predict how forest ecosystems will respond to future disturbances and global change processes.
Source: Soil Science Society of America