Scientists Solve Problem of Quantum Dot 'Blinking'
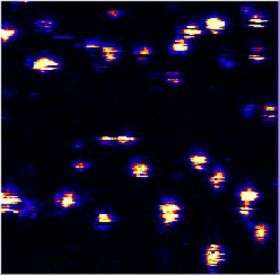
Quantum dots—tiny, intense, tunable sources of colorful light—are illuminating new opportunities in biomedical research, cryptography and other fields. But these semiconductor nanocrystals also have a secret problem, a kind of nervous tic. They mysteriously tend to “blink” on and off like Christmas tree lights, which can reduce their usefulness.
Scientists at JILA have found one possible way to solve the blinking problem and have induced quantum dots to emit photons (the smallest particles of light) faster and more consistently. The advance could make quantum dots more sensitive as fluorescent tags in biomedical tests and single-molecule studies and steadier sources of single photons for “unbreakable” quantum encryption. JILA is a joint venture of the National Institute of Standards and Technology and the University of Colorado at Boulder.
By bathing the dots in a watery solution of an antioxidant chemical used as a food additive, the JILA team increased photon emission rate four- to fivefold, a “shocking” result because the rate at which light radiates is generally considered an immutable property of the dot, JILA/NIST Fellow David Nesbitt says.
The JILA scientists dramatically reduced the average time delay between excitation of a quantum dot and resulting photon emission from 21 nanoseconds to 4 nanoseconds while reducing the probability of blinking up to 100 fold. Nesbitt calls blinking the “hidden dirty secret” of quantum dots. (Nesbitt notes that blinking is not always an annoyance. For example, it can serve as a measurement probe of very slow rates of electron flow through nanoscale materials).
The quantum dots used in the JILA experiments were made of cadmium-selenide cores just 4 nanometers wide coated with zinc sulfide. When a dot is excited by a brief laser pulse, one electron is separated from the “hole” it normally occupies.
A few nanoseconds later, the electron typically falls back into the hole, sometimes producing a single photon—always in a color that depends on dot size, greenish-yellow in this case. But every so often the electron fails to make it back to its hole and instead is ejected to imperfections on the dot’s surface. The chemical added at JILA apparently attaches to these imperfections, blocking the electron from being trapped and thereby preventing the dot from blinking off.
Citation: V. Fomenko and D. J. Nesbitt. Solution control of radiative and nonradiative lifetimes: a novel contribution to quantum dot blinking suppression. Nano Letters. Published online Dec. 21, 2007.
Source: National Institute of Standards and Technology