Researchers develop 'one-pot' porous surfaces for fuel cells

Cornell researchers have developed a "one-pot" process to create porous films of crystalline metal oxides that could lead to more-efficient fuel cells and solar cells.
In a fuel cell, a material with nanoscale pores offers more surface area over which a fuel can interact with a catalyst. Similarly in solar cells, a porous material offers more surface area over which light can be absorbed, so more of it is converted to electricity.
Previously such porous materials have been made on hard templates of carbon or silica, or by using soft polymers that self-assemble into a foamy structure. Making a hard porous template and getting the metal oxides to distribute evenly through it is tedious. The polymer approach is easier and makes a good structure, but the metal oxides must be heated to high temperatures to fully crystallize, and this causes the polymer pores to collapse.
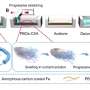
The Cornell researchers have combined what Ulrich Wiesner, Cornell professor of materials science and engineering, calls "the best of the two approaches," using a soft block copolymer called poly(isoprene-block-ethylene oxide) or PI-b-PEO that carbonizes when heated to high temperatures in an inert gas, providing a hard framework around which the metal oxide crystallizes. Subsequent heating in air burns away the carbon. Wiesner calls this "combined assembly by soft and hard chemistries," or CASH.
The research is described in an online paper in the journal Nature Materials by Wiesner, Francis DiSalvo, the J.A. Newman Professor of Chemistry and Chemical Biology, and colleagues.
The researchers created porous films of titanium oxide, used in solar cells, and niobium oxide, a potential fuel-cell catalyst support. Chemicals that will react to form the metal oxides and a solution of PI-b-PEO are combined. As the reaction proceeds, the PI portion of the copolymer forms cylinders some 20 nanometers across surrounded by metal oxides, and subsequent heat treatments leave uniform, highly crystalline metal oxide with cylindrical pores. The pores are neatly ordered in hexagonal patterns, which creates a larger surface area than if the pores were randomly distributed. "When the pores are ordered, you can get more of them into the same space," Wiesner explains.
The resulting materials were examined by electron microscopy, X-ray diffraction and a variety of other techniques, all of which confirmed a highly crystalline structure and a uniform porosity, the researchers reported.
The next step, Wiesner said, is to apply the CASH process to the creation of porous metals.
Co-authors of the Nature Materials paper are postdoctoral researcher Jinwoo Lee and graduate research assistants M. Christopher Orilall, Scott Warren and Marleen Kampeman.
Source: Cornell University