Nanotextured implant materials: blending in, not fighting back
Biomedical engineers are constantly coming up with ways to repair the human body, replacing defective and worn out parts with plastic, titanium, and ceramic substitutes – but the body does not always accept such substitutes seamlessly. Engineers from Brown and Purdue universities have found that simply changing the surface texture of implants can dramatically change the way cells colonize a wide variety of materials.
Two recent experiments have focused on the materials used in stents – those springy little cylinders that hold open once-clogged arteries – and artificial blood vessels. Currently only about 30 percent of small diameter blood vessel grafts (less than 6 mm diameter) last more than five years, and up to 20 percent of stents need to be replaced because the artery walls thicken in and around them in a process known as restenosis. Drug-coated stents were introduced years ago as one way to combat this problem, but concerns have surfaced recently about increased clotting.
Instead of using chemistry to fight the body's response to such foreign materials, Thomas Webster, an associate professor of engineering, and Karen Haberstroh, an assistant professor of engineering, thought maybe they could use physical structure to allow the foreign materials to blend in better. "What we're trying to do is fundamentally different," says Webster. "We're trying to find materials that the body accepts, rather than develop drugs or develop materials that will kill a cell – no matter if it kills a bad cell or a good cell. We're trying to find materials that accept good cells, as opposed to killing off bad cells."
Normal healthy blood vessels have a thin lining of specialized cells called the endothelium, surrounded by a thicker layer of smooth muscle cells that make up the arterial wall. The proteins collagen and elastin make up much of this lining and create a texture of fine nanoscale bumps on the inside of the blood vessel. This contrasts strongly with most of the materials used in implants, which have microscale texture, but are nearly smooth at the nanoscale.
When the researchers changed the surface texture of implant materials to better match the natural texture of the endothelium, they found that endothelial cells quickly colonized the foreign surfaces, effectively camouflaging them and preventing smooth muscle cells from overgrowing the implants. Once the endothelial cells form a single, solid layer, they stop piling on and switch to producing the proteins collagen and elastin.
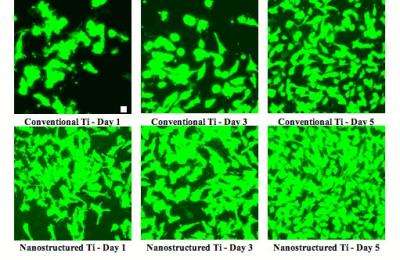
In one experiment, published with Purdue University graduate student Saba Choudhary in the journal Tissue Engineering, Webster and Haberstroh pressed together titanium particles that were less than 1 micron in size to create titanium with nanoscale surface texture. When they compared samples of the nanostructured material to conventional titanium in mixed cell culture, they found that the nanoscale surface features encouraged endothelial cells to colonize the material and spread much faster than smooth muscle cells. Where endothelial cells established themselves, they formed a single thin layer that inhibited overgrowth of the smooth muscle cells that tends to narrow stented arteries.
In another experiment, published in the Journal of Biomedical Materials Research with Purdue graduate student Derick Miller, the team molded pieces of PLGA, a biodegradable polymer often used for blood vessel grafts, so they came out completely covered with bumps that were 100, 200 or 500 nanometers in diameter. The surface with 200-nanometer features strongly favored the adsorption and spreading of fibronectin, a protein that helps endothelial cells quickly coat the graft.
Webster and Haberstroh's next step will be to test such nanostructured implants in live animals. If the same behavior holds true for materials placed in the body, the rapid growth of endothelial cells would help the implants to integrate quickly into existing blood vessels, provoking less immune response and a longer-lasting repair.
Source: Brown University