Nanoparticles for Delivery of Prostate Cancer Treatment
Alan Garen, professor of molecular biophysics and biochemistry at Yale has received a $100,000 award from the Prostate Cancer Foundation to expand research on the delivery of a targeted therapy for prostate cancer using nanoparticles.
Garen and his collaborator Zhiwei Hu have developed a way to directly target and destroy the blood vessels of solid tumors, thus destroying the tumors while leaving normal tissue unharmed.
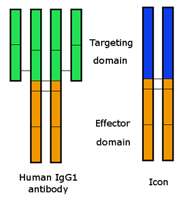
The technology uses a synthetic gene encoding an antibody-like molecule that activates an immune response to destroy the tumor blood vessels and associated tumors.
Garen previously received grants from the National Cancer Institute and Breast Cancer Alliance for Icon projects. The grant from the Prostate Cancer Foundation will extend his research to prostate cancer, which is the most common non-skin cancer in the United States.
Prostate cancer strikes one in six men. In 2007 alone, more than 200,000 men will be diagnosed with prostate cancer and more than 25,000 men will die of the disease. As the Baby Boomer generation begins to turn 60, increasing numbers of men are in the highest-risk sector for the disease. Over the next decade, the number of new cases is expected to increase to more than 300,000 annually.
The molecule that Garen and Hu constructed, called an Icon, recognizes the receptor tissue factor (TF) found on cells lining the inner surface of blood vessels in tumors but not in normal tissues. The Icon binds to TF more strongly and specifically than a natural antibody. Because the Icon acts through the blood, it can reach metastatic tumors throughout the body, which is critical for effective cancer therapy.
With the Prostate Foundation funding Garen will test the efficacy and safety of using targeted liposomal nanoparticle vectors — lipid-covered, gene delivery packets — to deliver the therapy in animal models of human metastatic prostate cancer.
“While we can directly inject the purified Icon molecule into the bloodstream, this procedure is less effective than having the Icon synthesized in vivo” said Garen. “We prefer to deliver the Icon gene to tumor cells, so that they cause their own destruction.”
The Yale scientists previously used a virus to deliver the Icon gene, a system that was effective and safe in animal models and is being prepared for a clinical trial.
“The advantage of nanoparticle vectors is that they do not reproduce, are not immunogenic, and are easier to produce than adenoviral vectors,” said Garen. The nanoparticles will have a tag on their outside that binds to the tumor blood vessels. After binding, they are taken up by the cells and unload the gene that codes for the Icon, allowing the cells to produce and secrete the Icon.
“The key is to have an efficient and safe way to deliver a specific and effective therapeutic agent,” said Garen. “Having the nanoparticle targeted specifically to tumor blood vessels, and the Icon derived entirely from human components, should enhance the safety and efficacy of the procedure.”
Source: Yale University