Researchers find a new way to read nanoscale vibrations
Nanomechanical oscillators -- tiny strips of vibrating silicon only a few hundred atoms thick -- are the subject of extensive study by nanotechnology researchers. They could someday replace bulky quartz crystals in electronic circuits or be used to detect and identify bacteria and viruses.
The catch is that measuring their vibrations isn't easy. It is usually done by bouncing laser beams off them -- which won't work when the nanodevices become smaller than the wavelength of the light -- or with piezoelectric devices -- those bulky quartz crystals we're trying to get rid of.
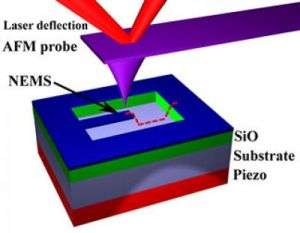
Now Cornell University researchers have come up with a very simple solution: reach out and touch them. The vibration of the tiny oscillators can be measured by "tapping" with an atomic force microscope (AFM).
An AFM uses a tiny probe that moves slowly just above a surface. Electrostatic attraction or repulsion between the atoms in the tip of the probe and those in the surface causes the probe to move up and down, creating an image of the surface so detailed that individual atoms show up as bumps. Alternatively, the AFM can be used in "tapping mode," literally bouncing off the surface.
"AFMs are all over the place," said Rob Ilic, research associate in the Cornell NanoScale Facility and lead author on a paper about the research published Feb. 23 in the online edition of the Journal of Applied Physics. "So this offers a simple way to study these structures." (Cornell, for example, has at least a dozen AFMs in various labs.) Moreover, he said, probes similar to those in an AFM can be built directly into nanofabricated devices.
This would amount to using MEMS to measure NEMS, he said. MEMS (microelectromechanical systems) are machines with moving parts measured in microns, or millionths of a meter; NEMS (nanoelectromechanical systems) are measured in nanometers, or billionths of a meter. A nanometer is about the length of three atoms in a row. When the NEMS oscillator is too small to be observed by laser light, it could still be coupled to a MEMS probe that in turn would be large enough for a laser readout.
To measure the vibration of a nanomechanical oscillator, the AFM probe moves along the length of the oscillating rod. The result is a complex bouncing interaction between the probe and the oscillator -- imagine shaking one end of a spring and watching the vibrations at the other end -- from which the frequency of vibration of the oscillator can be determined mathematically.
For the experiments just reported, Ilic and colleagues manufactured a wide variety of silicon cantilevers -- strips of silicon attached at one end with the other free to vibrate -- from 5 to 12 microns long, 1/2 to 1 micron wide and about 250 nanometers thick, which had natural vibration frequencies from 1 to 15 Mhz. The cantilevers were set into vibration by a piezoelectric device.
The experimenters first measured the resonant frequencies of the cantilevers by focusing laser beams on them and observing deflection of the reflected light, then scanned each cantilever with the AFM probe, both in tapping mode and with the probe just above the surface. They found the AFM measurements in good agreement with laser measurements, although the AFM readouts had a somewhat lower "quality factor," because the oscillator and probe were interacting. This would make the method somewhat less precise in mass detection.
Nanomechanical oscillators are often cited as potential tools for detecting bacteria, viruses or other organic molecules. An array of tiny cantilevers might be created with antibodies to many different pathogens attached to them. An experimental solution could then be washed over the array, allowing microbes to bind to the cantilevers with matching antibodies. Since the cantilevers are so tiny, an attached bacterium or virus represents a significant change in mass, which changes the frequency at which the oscillator will vibrate.
In a practical device, a MEMS probe could be mounted above each NEMS oscillator to read out which oscillators in the array show a change in frequency -- and thus identify which pathogens are present.
Source: Cornell University




















