Scientists develop rapid method for judging nanotube purity
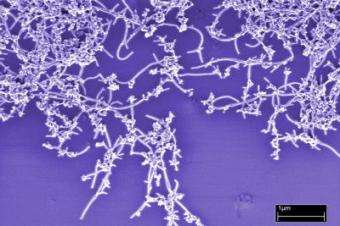
Researchers at the National Institute of Standards and Technology have developed a sensitive new method for rapidly assessing the quality of carbon nanotubes. Initial feasibility tests show that the method not only is faster than the standard analytic technique but also effectively screens much smaller samples for purity and consistency and better detects sample variability.
Carbon nanotubes have unique properties, and thermal and electrical conductance, that could be useful in fields such as aerospace, microelectronics and biotechnology. However, these properties may vary widely depending on nanotube dimensions, uniformity and chemical purity. Nanotube samples typically contain a significant percentage of more ordinary forms of carbon as well as metal particles left over from catalysts used in manufacturing.
The new NIST method, described at a conference last week, involves spraying nanotube coatings onto a quartz crystal, gradually heating the coated crystal, and measuring the change in its resonant frequency as different forms of carbon vaporize. The frequency changes in proportion to the mass of the coating, and scientists use this as a measure of stability at different temperatures to gauge consistency among samples. The quartz crystal technique, which can reveal mass changes of just a few nanograms, already is used in other contexts to detect toxic gases and measure molecular interactions.
NIST researchers tested dozens of samples from a batch of commercial single-walled carbon nanotubes, comparing results of the new method with those from a standard technique, thermogravimetric analysis, and confirming results with scanning electron microscopy. Both methods revealed that the samples contained large amounts of amorphous carbon as well as residual metal particles. But the quartz crystal method could obtain results from just micrograms of material, compared to milligrams for thermogravimetric analysis, and also revealed several orders of magnitude more variability in the samples tested. The new technique also uses simpler equipment.
Although the differences among nanotube samples may appear subtle, they may still affect product viability, because even small variations in material composition can affect electrical and thermal behavior, and lack of uniformity may demand higher loads of nanotubes, which are expensive. NIST scientists carried out the tests with the help of students from the University of Colorado-Boulder and Rensselaer Polytechnic Institute, Troy, N.Y.
Ref: S.A. Hooker, R. Geiss, R. Schilt and A. Kar. Rapid inspection of carbon nanotube quality. Paper presented Jan. 25 at the 31st International Cocoa Beach Conference and Exposition on Advanced Ceramics and Composites, Daytona Beach, Fla.
Source: National Institute of Standards and Technology





















