Nano probe may open new window into cell behavior
To create drugs capable of targeting some of the most devastating human diseases, scientists must first decode exactly how a cell or a group of cells communicates with other cells and reacts to a broad spectrum of complex biomolecules surrounding it.
But even the most sophisticated tools currently used for studying cell communications suffer from significant deficiencies. Typically, these tools can detect only a narrowly selected group of small molecules or, for a more sophisticated analysis, the cells must be destroyed for sample preparation. This process makes it very difficult to observe complex cellular interactions just as they would occur in their natural habitat -- the human body.
Georgia Tech researchers have created a nanoscale probe, the Scanning Mass Spectrometry (SMS) probe, that can capture both the biochemical makeup and topography of complex biological objects in their normal environment -- opening the door for discovery of new biomarkers and improved gene studies, leading to better disease diagnosis and drug design on the cellular level. The research was presented in the July issue of IEEE Electronics Letters.
The new instrument, a potentially very valuable tool for the emerging science of systems biology, may help researchers better understand cellular interactions at the most fundamental level, including cell signaling, as well as identifying protein expression and response to the external stimuli (e.g., exposure to drugs or changes in the environment) from the organ scale down to tissue and even the single cell level.
"At its core, disease is a disruption of normal cell signaling," said Dr. Andrei Fedorov, a professor in Georgia Tech's Woodruff School of Mechanical Engineering and lead researcher on the project. "So, if one understands the network and all signals on the most fundamental level, one would be able to control and correct them if needed. The SMS probe can help map all those complex and intricate cellular communication pathways by probing cell activities in the natural cellular environment."
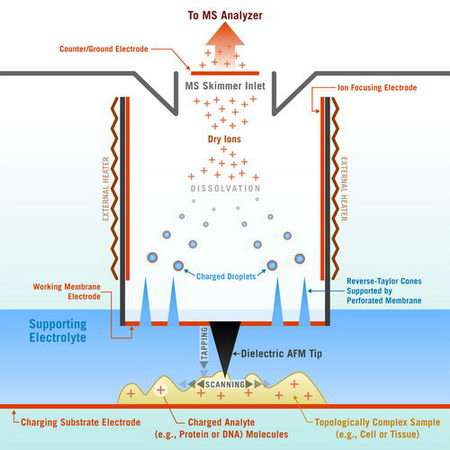
The SMS probe offers the capability to gently pull biomolecules (proteins, metabolites, peptides) precisely at a specific point on the cell/tissue surface, ionize these biomolecules and produce "dry" ions suitable for analysis and then transport those ions to the mass spectrometer (an instrument that can detect proteins present even in ultra-small concentrations by measuring the relative masses of ionized atoms and molecules) for identification. The probe does this dynamically (not statically), imaging the surface and mapping cellular activities and communication potentially in real time. In essence, in scanning mode, the SMS probe could create images similar to movies of cell biochemical activities with high spatial and temporal resolution.
The SMS probe can be readily integrated with the Atomic Force Microscope (AFM) or other scanning probes, and can not only image biochemical activity but also monitor the changes in the cell/tissue topology during the imaging.
"The probe potentially allows us to detect complex mechano-bio-electro-chemical events underlying cell communication, all at the same time!" Fedorov said. "The future work is in refinement of the idea and development of a versatile instrument that can be used by biological and medical scientists in advancing the frontiers of biomedical research."
The key challenge for the Georgia Tech team, which includes Dr. Levent Degertekin, was to create a way for a mass spectrometer, the primary tool for studying proteins, to sample biomolecules from a small domain and do it dynamically, thus enabling biochemical imaging. The researchers had to find a way to pull the targeted molecules out of the sample, as if they were using virtual tweezers, and then transfer these molecules into a dry and electrically charged state suitable for mass spectrometric analysis.
The solution to the problem came from a trick related to the basic fluid mechanics of ionic fluids, as the researchers exploited strong capillary forces to confine fluid within a nanoscale domain of the probe inlet (enabling natural separation of liquid and gaseous environments) and then used the classical Taylor electrohydrodynamic focusing of the jets to produce charged ions, but in reverse (pull) rather than in a commonly-used forward (push) mode.
Source: Georgia Institute of Technology





















