Brown Engineers Use DNA to Direct Nanowire Assembly and Growth
A small but growing number of engineers are using nature’s engineer – DNA – to create nanomaterials that can be used in everything from medical devices to computer circuits. A team from Brown University and Boston College is the first to use DNA to direct construction and growth of complex nanowires. Their work appears in Nanotechnology.
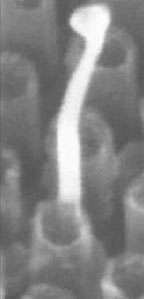
A research team led by Brown University engineers has harnessed the coding power of DNA to create zinc oxide nanowires on top of carbon nanotube tips. The feat, detailed in the journal Nanotechnology, marks the first time that DNA has been used to direct the assembly and growth of complex nanowires.
The tiny new structures can create and detect light and, with mechanical pressure, generate electricity. The wires’ optical and electrical properties would allow for a range of applications, from medical diagnostics and security sensors to fiber optical networks and computer circuits.
“The use of DNA to assemble nanomaterials is one of the first steps toward using biological molecules as a manufacturing tool,” said Adam Lazareck, a graduate student in Brown’s Division of Engineering. “If you want to make something, turn to Mother Nature. From skin to sea shells, remarkable structures are engineered using DNA.”
Lazareck, who works in the laboratory Jimmy Xu, professor of engineering and physics, led the research. The work is an example of “bottom up” nanoengineering. Instead of molding or etching materials into smaller components, such as computer circuits, engineers are experimenting with ways to get biological molecules to do their own assembly work. Under the right chemical conditions, molecular design and machinery – such as light-sensing proteins or viral motors – can be used to create miniscule devices and materials.
In this work, the team of engineers and scientists took the “bottom-up” approach one step further by successfully harnessing DNA to provide instructions for this self-assembly. The new structures created in the Xu lab are the first example of DNA-directed self-assembly and synthesis in nanomaterials.
The Xu lab is the first in the world to make uniform arrays of carbon nanotubes. Lazareck and his collaborators at Brown and Boston College built on this platform to make their structures. They started with arrays of billions of carbon nanotubes of the same diameter and height evenly spaced on a base of aluminum oxide film. On the tips of the tubes, they introduced a tiny DNA snippet.
This synthetic snippet of DNA carries a sequence of 15 “letters” of genetic code. It was chosen because it attracts only one complement – another sequence made up of a different string of 15 “letters” of genetic code. This second sequence was coupled with a gold nanoparticle, which acted as a chemical delivery system of sorts, bringing the complementary sequences of DNA together. To make the wires, the team put the arrays in a furnace set at 600° C and added zinc arsenide. What grew: Zinc oxide wires measuring about 100-200 nanometers in length.
The team conducted control experiments – introducing gold nanoparticles into the array with no DNA attached or using nanotubes with no DNA at the tips in the nanotube array – and found that very few DNA sequences stuck. And no wires could be made. Lazareck said the key is DNA hybridization, the process of bringing single, complimentary strands of DNA together to reform the double helices that DNA is famous for.
“DNA provides an unparalleled instruction manual because it is so specific,” Lazareck said. “Strands of DNA only join together with their complements. So with this biological specificity, you get manufacturing precision. The functional materials that result have attractive properties that can be applied in many ways.”
“We’re seeing the beginning of the next generation of nanomaterials,” said Xu, senior author of the article. “Many labs are experimenting with self-assembly. And they are making beautiful, but simple, structures. What’s been missing is a way to convey information – the instruction code – to make complex materials.”
Graduate student Teng-Fang Kuo was part of the Brown team along with two former graduate students in the Xu lab – Bradford Taft at Boston College and Sylvain Cloutier at the University of Delaware. Shana Kelley, professor of chemistry at Boston College, contributed to the experiments.
The Air Force Office of Scientific Research, the Office of Naval Research, the Defense Advanced Research Projects Agency and the National Sciences and Engineering Council of Canada funded the research.
Source: Brown University