July 1, 2006 feature
Electromechanical imaging in liquid environments: a pathway toward molecular-level resolution of biological systems
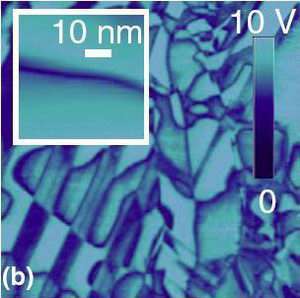
Scientists have demonstrated that electromechanical imaging techniques, when operated in a liquid environment, can provide a resolution of up to 10 times that of the resolution when imaging in air. The liquid imaging method will allow for high-res studies of ferroelectric materials, as well as imaging electromechanical properties of biological systems, ultimately on the molecular level.
Scientists from Oak Ridge National Laboratory in Tennessee have overcome several hurdles faced by electromechanical imaging techniques with a simple solution of operating piezoresponse force microscopy (PFM) in a liquid environment. The team found that imaging in an aqueous environment enabled the microscope to minimize negative effects, namely electrostatic tip-surface interactions and capillary forces, and overcome these barriers to achieve higher resolution imaging.
Physicists use electromechanical imaging to gain a better understanding of ferroelectric and piezoelectric materials, both of which exhibit unique electrical and mechanical properties. With PFM, scientists can probe the electromechanical properties of these materials in hopes of developing new technologies such as non-volatile random access memories, ultra high density data storage, and nanosensors and actuators.
“Higher resolution potentially enables us to gather more detailed information on the structure of domain walls and low dimensional ferroelectrics,” Sergei Kalinin, coauthor of the study in recent Physical Review Letters, told PhysOrg.com. “More importantly, imaging in solutions allows us to study the polarization-controlled chemical processes on ferroelectrics. Improvement in resolution brings us closer to the fundamental limits of ferroelectricity. The better you can resolve the domain wall, the better you can design and characterize materials with small domains. The smaller the domain, the higher the density of domains can be created, and thus, smaller, denser memory devices can be made.”
Ferroelectric materials exhibit pairs of opposite electrical charges – known as “dipole moments” – whose positive and negative areas can be reversed by applying an electric field. Piezoelectric materials such as tourmaline and quartz, which have been known for more than a hundred years to be able to generate electricity and light, can generate a voltage under application of mechanical stress (or, in the reverse process, can change their shape under application of voltage). These materials already provide many useful applications, from sonar to ultrasound, and scientists believe many more uses exist.
In their experiment, the scientists applied a high-frequency periodic voltage to a gold-coated tip of an atomic force microscope cantilever in contact with a piezoelectric material (all submerged in the liquid), thereby generating an electric field in material touched by the tip. The electric field deforms the surface of the piezoelectric material and displaces the microscope tip, registered through cantilever deflection. When measuring the surface topology, as well as the oscillating tip’s amplitude and phase, the scientists achieved extremely high resolution imaging. The team imaged widths of domain walls (which separate ferroelectric domains) as small as 3 nanometers, very close to the actual width of around 1 nanometer.
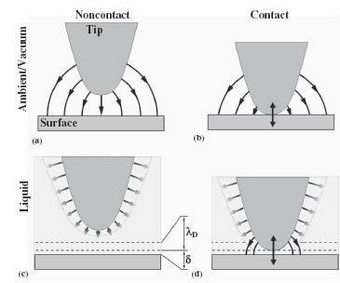
The increase in resolution, the scientists explain, stems from the fact that the liquid environment can eliminate electrostatic interactions between the tip and surface, which are detrimental to electromechanical imaging, at all but very small distances. Essentially, mobile ions in liquid screen electrostatic interactions, rendering them short-ranged and resulting in field localization in the tip-surface junction. Secondly, the liquid environment eliminates capillary interactions, which typically enlarge the tip-surface contact area and decrease resolution. The key to the successful imaging was the use of high excitation frequencies corresponding to high-order cantilever eigenmodes, minimizing viscous damping and added mass effects of liquid on cantilever dynamics, allowing for greater sensitivity.
“We were surprised that we could apply a bias to a tip in solution and still measure electromechanical response in a damped environment,” said Kalinin. “Also, the fact that mobile ions screen long-range interactions and make them effectively short range was an unexpected (rather, obvious in hindsight) discovery.”
Overcoming these hurdles enables electromechanical imaging to achieve a high resolution that has scientists speculating on a host of applications. One likely possibly is the human body, since piezoelectricity is linked to physiological structure.
“Piezoelectric coupling is a nearly-universal feature of biological systems, due to the combination of optical activity and polar bonding,” said Brian Rodriguez, coauthor of the study. “Biosystems involve complex forms of electromechanical coupling, such as ion channels, membrane flexoelectricity in outer hair cells and mitochondria, etc. Probing these electromechanical phenomena on the nano- and ultimately molecular scales will allow us to get insight into the fundamental mechanism for electromechnical and electrochemical transformations in biosystems. However, most of these systems can be studied only in liquid physiological environments.”
“Eventually, we hope that continued research in this direction will help to better understand how the electromechanical properties of biosystems are related to their functionality,” added Kalinin. “For example, understanding how the interplay between mechanical stress and applied bias on the nanoscale relates to problems such as stem cell differentiation into specific body cell types, bone regeneration and others.”
Citation: Rodriguez, Brian J., Jesse, Stephen, Baddorf, A.P. and Kalinin, Sergei V. “High Resolution Electromechanical Imaging of Ferroelectric Materials in a Liquid Environment by Piezoresponse Force Microscopy.” Physical Review Letters 96, 237602 (2006).
By Lisa Zyga, Copyright 2006 PhysOrg.com. All rights reserved. This material may not be published, broadcast, rewritten or redistributed.




















