Sticky synthetic molecules aid fight against prostate cancer
(PhysOrg.com) -- A team led by Yale University researchers has discovered a previously unknown binding site on a protein found on prostate cancer cells that can enhance their efforts to tag the cells for destruction.
The new site improves the ability of synthetic molecules, recently developed by the team, to attach to prostate cancer cells, with the ultimate goal of enhancing the body's immune response to the disease. Their findings appear online in the Journal of the American Chemical Society.
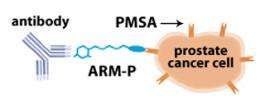
The new study builds on the team's recent development of a new class of synthetic molecules, called "antibody-recruiting molecules targeting prostate cancer" (ARM-Ps), which work by binding simultaneously to antibodies already present in the bloodstream and to a protein found on prostate cancer cells called prostate-specific membrane antigen (PSMA). By coating the cancer cells in antibodies, the ARM-P molecules flag them as a threat and trigger the body's own immune response, which does not recognize the cancer cells as foreign pathogens on its own.
While the researchers were developing different derivatives of the ARM-P molecules, they found that the molecule's length affected how strongly it bonded to the cancer cells. "We were surprised to find a ‘magic length' that maximized the affinity for the ARM-P molecules to bind to the prostate cancer cells," said David Spiegel, M.D., assistant professor of chemistry and lead author of the paper.
Spiegel and his colleagues realized that the ARM-P molecules, which were designed to bind to a single PSMA site, must have been binding to another site on the protein as well. Using advanced imaging techniques along with computational modeling, the team analyzed the previously unknown binding site and discovered that it is created by a single amino acid: tryptophan (the same chemical found in turkey that causes drowsiness).
"We're quite intrigued by the simplicity of this binding site and the possibility that similar binding sites could be common in other proteins," Spiegel said.
In the meantime, Spiegel and his team will work to exploit the new binding site on the PSMA protein to improve the binding between the ARM-P molecules and prostate cancer cells. Prostate cancer is the second leading cause of cancer-related death among American men, with one out of every six American men expected to develop the disease.
More information: Citation: DOI:10.1021/ja104591m
Provided by Yale University