Histone H1 regulates gene activity throughout the cell cycle
A protein that helps pack DNA into the cell nucleus has an important role in regulating gene activity, scientists report. The researchers found that the protein, histone H1, also takes part in the formation of ribosomes, the cellular workbenches on which all proteins are made.
The study appeared online May 3 in the Journal of Cell Biology.
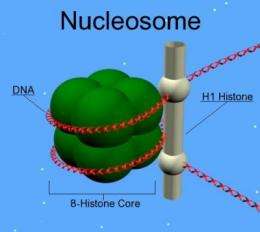
A human cell's genetic material is so vast that it must be condensed into tightly wound structures resembling beads on a string. The DNA winds around four core histone proteins to form one of the "beads" while H1 or "linker" histones clamp the DNA into place where it enters and exits the beads. One bead and its associated DNA make up a nucleosome. There are well over a million nucleosomes in the nucleus of a cell.
There are many varieties of the H1 histone protein in animals, making the histones difficult to study. Most research into histone biology has focused on the core histones, and previous studies have found that various cellular modifications of these other histones coincide with changes in gene activity.
The new study found that when H1 histones are modified by the addition of a phosphate group, a process called phosphorylation, that modification is associated with changes in gene activity in the vicinity of the phosphorylated histone.
"Most studies of histone phosphorylation have focused on cell division, when phosphorylation is at its peak," said Craig Mizzen, a professor of cell and developmental biology at the University of Illinois and corresponding author on the study. During cell division "much less of the genome is transcribed than at other points in the cell cycle," Mizzen said. "Everything is geared toward separating the replicated genome copies equally between the new daughter cells."
Suspecting that H1 phosphorylation was important for processes besides cell division, Mizzen and his colleagues identified the exact sites in H1 that are phosphorylated during various portions of the cell cycle.
Then-doctoral student Yupeng Zheng developed antibodies that recognize phosphorylation at the H1 sites cells use when they are not dividing. This enabled Zheng to discover that such "interphase" H1 phosphorylation is preferentially associated with genes when they are actually being transcribed.
"Histones are normally found all over the genome," Mizzen said, " and elucidating what is different about the nucleosomes on active genes versus those on repressed genes versus the rest of the genome, most of which is not protein-coding, is a central goal of current research in molecular biology."
Several core histone modifications are known to localize preferentially to active genes, Mizzen said. "But our work provides the first evidence that this is also true for H1 that is phosphorylated at specific sites."
Zheng also made a second surprising discovery. Using fluorescence microscopy to analyze cells, he noticed that the fluorescently labeled antibodies targeting phosphorylated H1 in non-dividing cells were lighting up the nucleolus, the region of the nucleus that is dedicated to transcribing ribosomal RNA, the special RNA upon which ribosomes are assembled.
"The ribosomal RNA genes are kept in the nucleolus and they're transcribed by a different enzyme system than the messenger RNAs that are transcribed from protein-coding genes," Mizzen said.
"The involvement of H1 phosphorylation in controlling ribosomal RNA gene transcription had not been suspected at all previously," Mizzen said. "That was a totally novel finding of our work."
Further experiments revealed that hormone treatments (in this case estradiol and glucocorticoid), stimulate the association of phosphorylated H1 histones with the regulatory regions of hormone-responsive genes.
The new findings could lead to a better understanding of alterations to the cell cycle associated with cancer and other diseases, Zheng said. Some drugs already are on the market that target kinases, the enzymes that phosphorylate other molecules, he said.
"One of the most successful drugs in leukemia treatment is a tyrosine kinase inhibitor, which inhibits a phosphorylation pathway and halts the growth of cancer cells," Zheng said.
Provided by University of Illinois at Urbana-Champaign
















