Advances reported in quest for drugs targeting childhood cancer
Investigators believe they have identified the founding member of a chemical family they hope will lead to a new class of cancer drugs, the first designed specifically against a childhood tumor, according to research led by St. Jude Children's Research Hospital scientists.
The chemical is the first small-molecule inhibitor to target the MDMX protein. Excess MDMX is a hallmark of the childhood eye tumor retinoblastoma as well as certain cases of breast, lung, prostate and other cancers. Nationally about 300 new cases of retinoblastoma are identified each year.
The discovery was reported online in advance of the April 2 print edition of the Journal of Biological Chemistry. An overabundance of MDMX or its sister protein, MDM2, can promote tumor progression by binding and suppressing a protein called p53. The role of p53 in normal cells is to induce death in cells that begin the unchecked cell division that is a hallmark of cancer.
MDM2 and MDMX use different mechanisms to disrupt the p53 pathway. There is an emerging scientific consensus that restoring normal p53 function might require shutting down both MDMX and MDM2. A small-molecule inhibitor against MDM2 is already in Phase 1 pharmaceutical industry trials. In this study, St. Jude researchers reported that when the new St. Jude compound, known as SJ-172550, is combined with an MDM2 inhibitor there was a corresponding increase in retinoblastoma cells death.
Michael Dyer, Ph.D., Developmental Neurobiology and the paper's senior author, said several years of detailed chemical studies and additional work are likely needed before SJ-172550 might be ready for human trials.
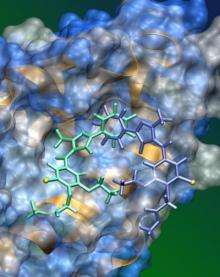
Evidence suggests SJ-172550 works by binding in a reversible manner to a pocket in the MDMX molecule. With SJ-172550 sitting in the pocket, the p53 protein cannot bind to MDMX, Dyer explained. That makes p53 available to do its job and eliminate tumor cells. About 65 percent of retinoblastoma tumors feature extra copies of the MDMX gene as do nearly 20 percent of patients with breast, colon and lung cancer.
The finding expands on work from Dyer's laboratory into the genetic and biochemical missteps that give rise to retinoblastoma. The advance reflects the combined efforts of the St. Jude departments of Developmental Neurobiology; Chemical Biology and Therapeutics; and Structural Biology. The first authors are Damon Reed, M.D., formerly of St. Jude and now of St. Petersburg, Fla., and Ying Shen, Ph.D., of Developmental Neurobiology.
"We went from a discovery in childhood cancer, MDMX amplification, to characterizing this first inhibitor in about three-and one-half years," Dyer said. "This model is now being replicated over and over in other cancers we treat at St. Jude."
Dyer's laboratory developed the biochemical and cell-based tests used by colleagues in Chemical Biology and Therapeutics. Researchers checked a chemical library of nearly 300,000 compounds using high-throughput screening. Investigators searched for molecules likely to block MDMX activity.
Dyer said the new compound might help researchers studying the biology of MDMX. "It may also be useful for any tumor that has normal p53," he said. "The idea is that if you have normal p53 and you need to turn it on, maybe by giving a drug that hits MDM2 and another that hits MDMX; you free p53 up to kill the cell."
Investigators' predictions of exactly how SJ-172550 interacts with MDMX are based on mathematical and computer models. Work is underway to capture an X-ray crystal structure of SJ-172550 bound to MDMX.
Provided by St. Jude Children's Research Hospital















