Newly explored bacteria reveal some huge RNA surprises
(PhysOrg.com) -- Yale University researchers have found very large RNA structures within previously unstudied bacteria that appear crucial to basic biological functions such as helping viruses infect cells or allowing genes to "jump" to different parts of the chromosome.
These exceptionally large RNA molecules have been discovered using DNA sequence data available within the past few years. The findings, reported in the December 3 issue of the journal Nature, suggest many other unusual RNAs remain to be found as researchers explore the genes of more species of bacteria, said Ronald Breaker, senior author of the paper and professor of Molecular, Cellular and Developmental Biology.
"Our work reveals new classes of large RNAs exist, which would be akin to protein scientists finding new classes of enzymes," said Breaker, a Howard Hughes Medical Institute investigator. "Since we have only scratched the surface when it comes to examining microbial DNA that is covering the planet - there will certainly be many more large RNAs out there to discover and these newfound RNAs are also likely to have amazing functions as well."
The RNA molecules rank among the largest and most sophisticated RNAs yet discovered and may act like enzymes or carry out other complex functions in bacteria. The RNAs are found in bacteria which have yet to be grown in labs and so have been difficult to study.
RNA, or ribonucleic acid, is a chemical related to DNA. (Move definition up) RNA molecules are best known for carrying information from genes encoded in DNA to ribosomes, which are the protein-manufacturing machines of cells. However, some RNAs are not passive messengers, but form intricate structures that function like enzymes. For example, ribosomes are constructed using the two largest structured RNAs in bacteria that together function as the chemical factory for producing proteins. Yale University's Thomas Steitz won the 2009 Nobel Prize for his work to solve the atomic-resolution structure of ribosomes from bacterial cells. His work helped prove that ribosomes stitch together amino acids to make proteins using large RNAs like enzymes.
Nearly all of the largest structured RNAs previously known had been discovered in the 1970s or earlier. The scientists discovered these new RNAs by analyzing genetic data from poorly studied bacteria that in many cases cannot yet be grown in laboratory conditions. Only a tiny fraction of bacteria in the wild can now be grown in the lab, and scientists have only recently been able to collect genetic data from uncultivated bacteria. Consequently, there is a vast array of bacteria for which genetic data remains unavailable. Many other RNAs likely remain hidden in these under-studied bacteria that also have unusual characteristics that will greatly expand the known roles of RNA in biology.
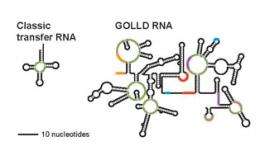
The Breaker laboratory has used the explosion of DNA sequence information and new computer programs to discover six of the top twelve largest bacterial RNAs just in the last several years. One of the newly discovered RNAs, called GOLLD, is the third largest and most complex RNA discovered to date, and appears to be used by viruses that infect bacteria. Another large RNA revealed in the study, called HEARO, has a genetic structure that suggests it is part of a type of "jumping gene" that can move to new locations in the bacterial chromosome. They also found other RNAs in species of bacteria abundant in the open ocean, and some of these had been identified near Hawaii by researchers from the Massachusetts Institute of Technology. These RNAs are also very common in bacteria that live near the shore of the North American east coast, and so organisms that carry this RNA are likely to be very common in the waters of all the earth's oceans.
More information: The authors of this paper are Zasha Weinberg, Jonathan Perreault, Michelle M. Meyer and Ronald R. Breaker. All are from Yale. The title of the paper is "Exceptional Structured Noncoding RNAs Revealed by Bacterial Metagenome Analysis."
Source: Yale University (news : web)

















