Magnetic Nanoworms and Nanocrystals Deliver siRNA to Tumors
(PhysOrg.com) -- Small pieces of nucleic acid known as short interfering RNAs, or siRNAs, can turn off the production of specific proteins, a property that makes them one of the more promising new classes of anticancer drugs in development. Indeed, at least two siRNA-based anticancer therapies, both delivered to tumors in nanoparticles, have begun human clinical trials.
Now, investigators at the Massachusetts Institute of Technology (MIT) have developed a modular nanoparticle-based drug delivery system that maximizes the quantity of siRNA molecules that not only can enter cells but also can escape into the cytoplasm, where they can interfere with protein production. Sangeeta Bhatia, M.D., Ph.D., and Phillip A. Sharp, Ph.D., of the MIT-Harvard Center of Cancer Nanotechnology Excellence, and Alain Charest, Ph.D., M.Sc., Tufts University School of Medicine, led the study, whose results appear in the journal ACS Nano.
The new siRNA delivery vehicle is made of dendrimer-conjugated magnetic and fluorescent nanoworms that the investigators call “dendriworms.” Dendrimers are synthetic polymers that generally have a spherical shape and that can be readily modified to carry a wide range of molecules, including nucleic acids. In this work, Dr. Bhatia and her colleagues used polyamidoamine dendrimers, which a large body of preclinical work has shown are fully biocompatible, and coupled them to a chain of magnetic nanoparticles known as a nanoworm. The investigators also added a fluorescent molecule to the nanoworms, thereby creating a drug delivery vehicle that they could also image in the body using either magnetic resonance imaging or fluorescence imaging. In a final step, the researchers added siRNA to the dendriworms. The resulting construct, which contains approximately 7 magnetic nanoparticles, 45-50 dendrimers, and 50 siRNA molecules, was stable under test conditions for up to 6 hours.
When added to cells growing in culture, this siRNA-dendrimer complex rapidly entered the cells and then escaped into the cellular cytoplasm. The researchers observed no significant toxicity in these in vitro experiments. When the dendriworms were administered to human glioblastoma cells, the delivered siRNA was able to silence production of the targeted gene, in this case a mutant gene known to be involved in glioblastoma development.
To test whether this dendriworm would work in a living animal, the researchers used a strain of mice that were genetically engineered to develop glioblastoma tumors spontaneously in the brain. The investigators found that the dendriworms were able to penetrate the tumors, deliver their therapeutic siRNA cargo into tumor cells, and silence the targeted gene in those cells.
Meanwhile, a group of investigators at The Jikei University School of Medicine in Tokyo, Japan, led by Yoshihisa Namiki, M.D., has demonstrated that lipid-coated magnetic crystals can safely and effectively deliver therapeutic siRNA to tumors in mice. The results of their experiments were published in the journal Nature Nanotechnology.
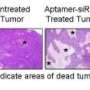
Dr. Namiki and his colleagues created their delivery vehicle by coating iron oxide nanocrystals with a layer of positively charged lipids. This layer binds strongly to siRNA molecules, which— like all nucleic acids—have a strong negative charge. After optimizing the lipid coating to maximize siRNA delivery efficiency, the investigators used their nanoparticle to deliver an anti-EGFR siRNA to gastric tumors in mice. After injecting the mice with the therapeutic nanoparticles, the investigators applied a local magnetic field around the vicinity of the tumor. After 28 days, tumors in the treated mice were 50% smaller compared with tumors in mice treated with just the nanoparticle and no siRNA.
The work with dendriworms, which is detailed in the paper “Functional delivery of siRNA in mice using dendriworms,” was supported by the NCI Alliance for Nanotechnology in Cancer, a comprehensive initiative designed to accelerate the application of nanotechnology to the prevention, diagnosis, and treatment of cancer. Investigators from Brigham and Women’s Hospital also participated in this study. An abstract is available at the journal’s Web site.
The work on magnetically guided siRNA therapy is detailed in the paper “A novel magnetic crystal-lipid nanostructure for magnetically guided in vivo gene delivery.” An abstract of this paper is available at the journal’s Web site.
Provided by National Cancer Institute (news : web)