New class of compounds discovered for potential Alzheimer's disease drug
A new class of molecules capable of blocking the formation of specific protein clumps that are believed to contribute to Alzheimer's disease pathology has been discovered by researchers at the University of Pennsylvania School of Medicine. By assaying close to 300,000 compounds, they have identified drug-like inhibitors of AD tau protein clumping, as reported in the journal Biochemistry.
Co-authors Alex Crowe, Research Specialist; Kurt R. Brunden, PhD, Director of Drug Discovery at Penn's Center for Neurodegenerative Disease Research (CNDR); Virginia M.-Y. Lee, PhD, and John Q. Trojanowski, MD, PhD, CNDR Co-Directors, and colleagues conducted the screen to find small molecules that prevent the formation of the tau protein fibrils. These fibrils, a hallmark pathological feature of AD, have been a holy grail for investigators hoping to better treat AD and related neurodegenerative diseases.
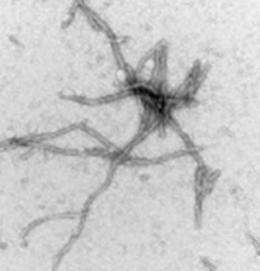
Tau fibrils accumulate as insoluble deposits in brain nerve cells of patients with a host of debilitating neurodegenerative diseases, the most prevalent of which is AD. Since these tau aggregates are found in several neurodegenerative disorders and are thought to contribute to disease pathology, it is hoped that drugs that prevent these deposits might prove to be effective therapeutic agents for AD and related disorders. This is the largest drug screen completed to date using the compound repository housed at the National Institutes of Health (NIH) Chemical Genomics Center.
Small molecules that prevent or reverse tau clumping might have therapeutic value, explains Brunden. Examples of such compounds have been described previously but nearly all have properties, such as chemical reactivity, poor absorption by the gut, or poor brain penetration, that render them unsuitable as drug candidates.
The test-tube-based assay used to screen the large NIH library was designed by Dr. Lee to see if each compound could block fibril formation in a sample of purified tau protein. Fluorescent tags embedded in the tau protein signal fibril formation as well as intermediary protein structures on the way to becoming fully formed fibrils. Fibril formation was also monitored with a specific dye that can bind to tau fibrils but not normal tau.
The team found a total of 285 compounds that were of potential interest, and of these they focused on a specific chemical series called ATPZs that effectively block fibril formation. The ATPZs fit most of the criteria for potential drug candidacy such as proper size, desirable chemical properties, specificity for the tau protein, and a predicted likelihood of crossing the blood-brain barrier. "This led us to further explore this unique chemical series," says Brunden.
They and NIH colleagues made or obtained 21 ATPZ analogue compounds, each with a similar core structure but having different chemical side-chains. "Think of this as variations on a theme," says Brunden "Certain variations will lead to improved compound potency compared to others and we measured this by how well each ATPZ analogue inhibited tau fibril formation."
By comparing how various side-chain changes affect the properties of the ATPZs, knowledge is gained on how to better improve compounds. Although increasing the potency of compounds is an important part of the process of developing a drug candidate, other aspects of compounds such as residence time in the body, the ability to be absorbed after oral administration, brain penetration and overall safety must all be considered during this optimization process.
"While we are excited about the discovery of this new series of tau fibril inhibitors, we are still a long ways from turning these early lead compounds into drugs," Brunden cautions. "However, we believe that certain of our ATPZ compounds will be very useful in allowing us to gain a better understanding of the consequences of inhibiting tau fibril formation in transgenic mouse models of Alzheimer's disease."
Source: University of Pennsylvania School of Medicine (news : web)


















