Researchers Suggest New Approach in Development Efforts for Parkinson’s Therapeutics
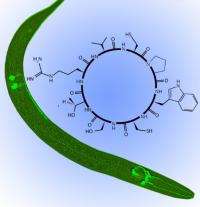
(PhysOrg.com) -- Researchers outline today a new approach in the potential development of drugs to counter a cellular defect that triggers Parkinson’s and other diseases.
A research article, publishing in this week’s Nature Chemical Biology, describes new avenues that protect, within model organisms, against the harmful clumping of proteins implicated in Parkinson’s and other neurodegenerative diseases, such as Alzheimer’s and Huntington’s. Three University of Alabama scientists are among the article’s co-authors.
“It expands the toolbox that exists for drug discovery,” said Dr. Guy Caldwell, associate professor of biological sciences at UA, of the research.
The researchers identified distinct cyclic peptides - amino acids linked in a ringlet - that protect against the problems resulting from the presence of extra copies of a protein, alpha synuclein.
Proteins must fold properly within cells. When extra copies of alpha synuclein are present, a series of misfoldings can occur, leading to aggregation, or clumping, of proteins. Such protein aggregation within the brain’s dopamine-producing neurons can lead to their malfunction or cell death, triggering the symptoms of Parkinson’s.
Shusei Hamamichi, a doctoral student, was UA’s lead researcher on the paper and was joined at UA by Caldwell and Dr. Kim Caldwell, assistant professor of biological sciences, in the research. The multi-institution effort, led by Dr. Susan Lindquist, of the Whitehead Institute and M.I.T., involved researchers representing five different universities and research institutes.
Cyclic peptides, which are naturally occurring in lower organisms, such as bacteria, have not been found in humans, Hamamichi said. They have routinely been made in test tubes, and some antibiotics are developed from them.
“What’s different here,” Caldwell said, “is the peptide is being made in vivo - inside an animal or inside a cell. There is an enzymatic reaction that occurs in the cell that cyclizes it in a living organism.”
The researchers used both yeast cells and tiny nematodes, known as C. elegans, as model organisms in their research.
“The key is the cyclic peptide is actually coded for genetically. It’s coded for by the DNA that we introduce into the yeast cell or worm neurons.”
Through analysis of some 5 million possibilities, the researchers identified specific cyclic peptides that, when modified with enzymes, provided the desired protective capabilities.
Using the cyclic peptides in this manner, as well as the strategy followed in screening them, represent novel approaches, Caldwell said.
A next step, Caldwell said, would be to test the possibilities within a mammal, such as a rodent. The long-range goal would be to develop a drug from the cyclic peptide that would offer its protection.
“This offers a new family of therapeutic agents that have not previously been applied to neurodegenerative diseases, specifically Parkinson’s,” he said.
Hamamichi, who in April won the UA graduate school’s Excellence in Research by a Doctoral Student award, is scheduled to earn his doctorate from UA in December and has accepted a post-doctoral position in Lindquist’s Whitehead lab for 2010.
Provided by University of Alabama















