May 5, 2009 weblog
Scientists Measure Differences Between Normal and Cancer Cell Surfaces
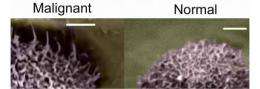
(PhysOrg.com) -- Scientists know that cancerous cells and normal cells have different physical features, but the details of these differences, and why they occur, are not well understood. In a recent edition of Nature Nanotechnology, researchers report measurements of certain physical differences between the surfaces of normal and cancerous cells, suggesting a new way to characterize cancer cells and a possible route for detection.
The group, composed of researchers from the Nanoengineering and Biotechnology Laboratories Center at Clarkson University, was studying human cervical cells. Led by Igor Sokolov, they focused on the cells' surface features, including microridges and hair-like microvilii, which, perhaps acting like sensors, are one key way that the cells interact with their environment. Together, these features form a cell's "brush."
They found that normal cervical cells tend to have a brush layer consisting of a single average length - 2.4 micrometers (millionths of a meter) - while the cancerous cells have mostly two typical lengths - 2.6 and 0.45 micrometers. Additionally, their analysis showed that the long cancer-cell brush is about half as dense as that of the normal-cell brush while the short cancer-cell brush is more than twice as dense.
The group made these findings using an atomic force microscope (AFM), a high-resolution device that can resolve details down to a fraction of a nanometer. The AFM works by scanning a surface with a tiny cantilever, a beam supported on one end so that it can move up and down. In an AFM, the beam is tipped with a nanometer-scale curved needle often made of silicon or silicon nitride. When brought near a sample, forces between the needle tip and the surface cause the cantilever to deflect. When the entire surface is scanned, the result is a set of force data that represents a surface map of the sample. By analyzing the forces, researchers can recover the nature and type of surface interactions.
In previous studies, scientists treated the surface of a cell as flat. In their work, the Clarkson researchers used various supporting techniques, including electron microscopy and confocal scanning laser microscopy, to show that the cell surface is sufficiently "brushy" to be visible in the AFM data. The researchers processed the forces using a "brush on soft surface" model, the type of model used to study polymer brushes (polymer chains tethered to a surface). Prior to this work, scientists had not looked at cell brush in this way.
The AFM method has an edge over other microscopy techniques, such as electron microscopy, because it can work with viable cells, avoiding misrepresentations of the cell structure and saving time on sample preparation.
More information: Nature Nanotechnology advance online publication 12 April 2009, DOI:10.1038/nnano.2009.77
© 2009 PhysOrg.com
















