Nanotech coating could lead to better brain implants to treat diseases
(PhysOrg.com) -- Biomedical and materials engineers at the University of Michigan have developed a nanotech coating for brain implants that helps the devices operate longer and could improve treatment for deafness, paralysis, blindness, epilepsy and Parkinson's disease.
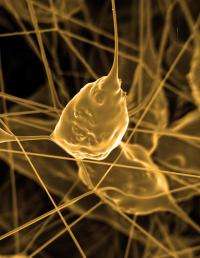
Currently, brain implants can treat Parkinson's disease, depression and epilepsy. These and the next generation of the devices operate in one of two ways. Either they stimulate neurons with electrical impulses to override the brain's own signals, or they record what working neurons are transmitting to non-working parts of the brain and reroute that signal.
On-scalp and brain-surface electrodes are giving way to brain-penetrating microelectrodes that can communicate with individual neurons, offering hope for more precise control of signals.
In recent years, researchers at other institutions have demonstrated that these implanted microelectrodes can let a paralyzed person use thought to control a computer mouse and move a wheelchair. Michigan researchers' say their coating can most immediately improve this type of microelectrode.
Mohammad Reza Abidian, a post-doctoral researcher in the Department of Biomedical Engineering who is among the developers of the new coating, says the reliability of today's brain-penetrating microelectrodes often begins to decline after they're in place for only a few months.
"You want to be able to use these for at least a couple years," Abidian said. "Current technology doesn't allow this in most cases because of how the tissues of the brain respond to the implants. The goal is to increase their efficiency and their lifespans."
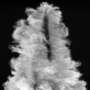
The new coating Abidian and his colleagues developed is made of three components that together allow electrodes to interface more smoothly with the brain. The coating is made of a special electrically-conductive nanoscale polymer called PEDOT; a natural, gel-like buffer called alginate hydrogel; and biodegradable nanofibers loaded with a controlled-release anti-inflammatory drug.
The PEDOT in the coating enables the electrodes to operate with less electrical resistance than current models, which means they can communicate more clearly with individual neurons.
The alginate hydrogel, partially derived from algae, gives the electrodes mechanical properties more similar to actual brain tissue than the current technology. That means coated neural electrodes would cause less tissue damage.
The biodegradable, drug-loaded nanofibers fight the "encapsulation" that occurs when the immune system tells the body to envelop foreign materials. Encapsulation is another reason these electrodes can stop functioning properly. The nanofibers fight this response well because they work with the alginate hydrogel to release the anti-inflammatory drugs in a controlled, sustained fashion as the nanofibers themselves break down.
"Penetrating microelectrodes provide a means to record from individual neurons, and in doing so, there is the potential to record extremely precise information about a movement or an intended movement. The open question in our field is what is the trade-off: How much invasiveness can be tolerated in exchange for more precision?" said Daryl Kipke, a professor in the Department of Biomedical Engineering and the director of the U-M Center for Neural Communication Technology.
In these experiments, the Michigan researchers applied their coating to microelectrodes provided by the U-M Center for Neural Communication Technology.
More information: A paper on this research, called "Multifunctional Nanobiomaterials for Neural Interfaces," is published in Advanced Functional Materials. It is the cover story on the February 24 issue.
Source: University of Michigan